The boiling points of hex-1-ene (64 °C) and hex-1-yne (71 °C) are sufficiently close that it is difficult to achieve a clean separation by distillation. Show how you might use the acidity of hex-1-yne to remove the last trace of it from a sample of hex-1-ene.

Show how you might synthesize the following compounds, using acetylene and any suitable alkyl halides as your starting materials. If the compound given cannot be synthesized by this method, explain why.
a. hex-1-yne
b. hex-2-yne
c. hex-3-yne
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Alkyne Synthesis
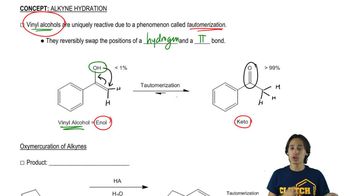
Markovnikov's Rule

Rearrangement and Isomerization
Solved Problem 9-1 showed the synthesis of dec-3-yne by adding the hexyl group first, then the ethyl group. Show the reagents and intermediates involved in the other order of synthesis of dec-3-yne, by adding the ethyl group first and the hexyl group last.
Show how you might synthesize the following compounds, using acetylene and any suitable alkyl halides as your starting materials. If the compound given cannot be synthesized by this method, explain why.
d. 4-methylhex-2-yne
e. 5-methylhex-2-yne
f. cyclodecyne
Show how you would synthesize each compound, beginning with acetylene and any necessary additional reagents
(a)
Show how you would synthesize each compound, beginning with acetylene and any necessary additional reagents
(b)
Predict the products of the following acid–base reactions, or indicate if no significant reaction would take place.
a. H—C≡C—H + NaNH2
b. H—C≡C—H + CH3Li
c. H—C≡C—H + NaOCH3
