- 1. The Chemical World9m
- 2. Measurement and Problem Solving2h 19m
- 3. Matter and Energy2h 15m
- Classification of Matter18m
- States of Matter8m
- Physical & Chemical Changes19m
- Chemical Properties8m
- Physical Properties5m
- Temperature (Simplified)9m
- Law of Conservation of Mass5m
- Nature of Energy5m
- First Law of Thermodynamics7m
- Endothermic & Exothermic Reactions7m
- Heat Capacity17m
- Thermal Equilibrium (Simplified)8m
- Intensive vs. Extensive Properties13m
- 4. Atoms and Elements2h 33m
- The Atom (Simplified)9m
- Subatomic Particles (Simplified)11m
- Isotopes17m
- Ions (Simplified)22m
- Atomic Mass (Simplified)17m
- Periodic Table: Element Symbols6m
- Periodic Table: Classifications11m
- Periodic Table: Group Names8m
- Periodic Table: Representative Elements & Transition Metals7m
- Periodic Table: Phases (Simplified)8m
- Periodic Table: Main Group Element Charges12m
- Atomic Theory9m
- Rutherford Gold Foil Experiment9m
- 5. Molecules and Compounds1h 50m
- Law of Definite Proportions9m
- Periodic Table: Elemental Forms (Simplified)6m
- Naming Monoatomic Cations6m
- Naming Monoatomic Anions5m
- Polyatomic Ions25m
- Naming Ionic Compounds11m
- Writing Formula Units of Ionic Compounds7m
- Naming Acids18m
- Naming Binary Molecular Compounds6m
- Molecular Models4m
- Calculating Molar Mass9m
- 6. Chemical Composition1h 23m
- 7. Chemical Reactions1h 43m
- 8. Quantities in Chemical Reactions1h 8m
- 9. Electrons in Atoms and the Periodic Table2h 32m
- Wavelength and Frequency (Simplified)5m
- Electromagnetic Spectrum (Simplified)11m
- Bohr Model (Simplified)9m
- Emission Spectrum (Simplified)3m
- Electronic Structure4m
- Electronic Structure: Shells5m
- Electronic Structure: Subshells4m
- Electronic Structure: Orbitals11m
- Electronic Structure: Electron Spin3m
- Electronic Structure: Number of Electrons4m
- The Electron Configuration (Simplified)20m
- The Electron Configuration: Condensed4m
- Ions and the Octet Rule9m
- Valence Electrons of Elements (Simplified)5m
- Periodic Trend: Metallic Character4m
- Periodic Trend: Atomic Radius (Simplified)7m
- Periodic Trend: Ionization Energy (Simplified)9m
- Periodic Trend: Electron Affinity (Simplified)7m
- Electron Arrangements5m
- The Electron Configuration: Exceptions (Simplified)12m
- 10. Chemical Bonding2h 10m
- Lewis Dot Symbols (Simplified)7m
- Ionic Bonding6m
- Covalent Bonds6m
- Lewis Dot Structures: Neutral Compounds (Simplified)8m
- Bonding Preferences6m
- Multiple Bonds4m
- Lewis Dot Structures: Multiple Bonds10m
- Lewis Dot Structures: Ions (Simplified)8m
- Lewis Dot Structures: Exceptions (Simplified)12m
- Resonance Structures (Simplified)5m
- Valence Shell Electron Pair Repulsion Theory (Simplified)4m
- Electron Geometry (Simplified)7m
- Molecular Geometry (Simplified)9m
- Bond Angles (Simplified)11m
- Dipole Moment (Simplified)14m
- Molecular Polarity (Simplified)7m
- 11 Gases2h 7m
- 12. Liquids, Solids, and Intermolecular Forces1h 11m
- 13. Solutions3h 1m
- 14. Acids and Bases2h 14m
- 15. Chemical Equilibrium1h 27m
- 16. Oxidation and Reduction1h 33m
- 17. Radioactivity and Nuclear Chemistry53m
Complete Ionic Equations: Videos & Practice Problems
Complete Ionic Equations are written from a molecular equation by showing each aqueous compounds as separate ions, while substances in the solid, liquid, or gas state remain intact. This means only soluble compounds break apart, so using solubility rules is essential for deciding what stays aqueous and what forms a precipitate. When an aqueous ionic compound is separated into ions, the coefficient must be distributed to give the correct number of each ion.
The usual progression is molecular equation \\(\rightarrow\\) complete ionic equation \\(\rightarrow\\) net ionic equation . In the complete ionic equation, all dissolved ions are shown explicitly. From there, spectator ions are identified as ions that appear unchanged on both sides of the equation and are removed to produce the net ionic equation. This helps focus on the ions that actually participate in the chemical reaction.
Complete Ionic Equations show aqueous compounds as fully dissociated ions.
Complete Ionic Equations
Complete Ionic Equations
Complete Ionic Equations Video Summary

The complete ionic equation shows all the aqueous compounds broken up into ions.
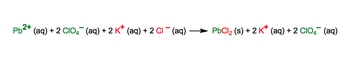
Complete Ionic Equations Example 1
Complete Ionic Equations Example 1 Video Summary
To convert a molecular equation into a complete ionic equation, it is essential to recognize which compounds can dissociate into ions. In this case, we have the reaction of 3 moles of calcium bromide (CaBr2) aqueous with 2 moles of lithium phosphate (Li3PO4) aqueous, resulting in the formation of 6 moles of lithium bromide (LiBr) aqueous and 1 mole of calcium phosphate (Ca3(PO4)2) solid.
Only the aqueous compounds will dissociate into their respective ions. Therefore, we will break down the calcium bromide, lithium phosphate, and lithium bromide into their ionic forms, while the calcium phosphate remains intact as a solid.
Starting with calcium bromide, the dissociation can be represented as follows:
3 CaBr2 (aq) → 3 Ca2+ (aq) + 6 Br- (aq)
Next, for lithium phosphate:
2 Li3PO4 (aq) → 6 Li+ (aq) + 2 PO43- (aq)
Finally, lithium bromide dissociates as:
6 LiBr (aq) → 6 Li+ (aq) + 6 Br- (aq)
Since calcium phosphate is a solid, it does not dissociate:
1 Ca3(PO4)2 (s)
Combining all these components, the complete ionic equation is:
3 Ca2+ (aq) + 6 Br- (aq) + 6 Li+ (aq) + 2 PO43- (aq) → 6 Li+ (aq) + 6 Br- (aq) + 1 Ca3(PO4)2 (s)
In summary, when converting to a complete ionic equation, remember to only break apart aqueous compounds and distribute coefficients to the respective ions formed.
Complete Ionic Equations
Complete Ionic Equations Video Summary
A net ionic equation is a simplified representation of a chemical reaction that highlights the ions directly involved in the reaction while omitting the spectator ions. Spectator ions are those that appear unchanged on both sides of the equation, meaning they do not participate in the actual chemical change. To derive a net ionic equation, one must first start with the molecular equation, which represents the reactants and products in their molecular form.
From the molecular equation, the next step is to write the complete ionic equation. This equation breaks down all soluble ionic compounds into their respective ions, showing all species present in the reaction. Finally, by removing the spectator ions from the complete ionic equation, we arrive at the net ionic equation, which succinctly illustrates the essential chemical changes occurring during the reaction.
This process of transitioning from a molecular equation to a net ionic equation is crucial for understanding the specific interactions between ions in a solution, allowing for a clearer insight into the underlying chemistry of the reaction.
Net Ionic Equation shows only the ions participating in the chemical reaction, without the spectator ions.
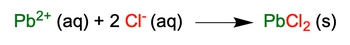
Complete Ionic Equations Example 2
Complete Ionic Equations Example 2 Video Summary
When ammonium sulfate reacts with calcium chloride, the first step is to write the molecular equation. The reactants can be represented as ammonium sulfate (NH4)2SO4 and calcium chloride CaCl2. The balanced molecular equation for this reaction is:
(NH4)2SO4 (aq) + CaCl2 (aq) → 2 NH4Cl (aq) + CaSO4 (s)
In this equation, ammonium sulfate and calcium chloride are both soluble in water, while calcium sulfate precipitates as a solid due to its low solubility.
Next, we break down the soluble compounds into their ionic forms to create the complete ionic equation. Ammonium sulfate dissociates into 2 ammonium ions (2 NH4+) and 1 sulfate ion (SO42-), while calcium chloride dissociates into 1 calcium ion (Ca2+) and 2 chloride ions (2 Cl-). The complete ionic equation is:
2 NH4+ (aq) + SO42- (aq) + Ca2+ (aq) + 2 Cl- (aq) → 2 NH4+ (aq) + 2 Cl- (aq) + CaSO4 (s)
In this equation, the ammonium ions and chloride ions are spectator ions, as they appear on both sides of the equation. To derive the net ionic equation, we remove these spectator ions, leaving us with:
SO42- (aq) + Ca2+ (aq) → CaSO4 (s)
This net ionic equation highlights the essential chemical change occurring in the reaction, which is the formation of solid calcium sulfate from the sulfate and calcium ions in solution. Understanding these steps is crucial for mastering the concepts of molecular, complete ionic, and net ionic equations in chemical reactions.
Provide the net ionic equation that occurs when the following aqueous compounds are mixed together:
Copper (II) Bromide and Lithium Hydroxide
Which of the following reagents could be used to separate the two anions from a solution containing magnesium nitrate and cesium hydroxide?
Which of the following reagents could be used to separate the two cations from a solution containing Lead (IV) acetate and cesium permanganate?
Do you want more practice?
More setsGo over this topic definitions with flashcards
More setsHere's what students ask on this topic:
A complete ionic equation represents all aqueous compounds as their individual ions, while solids, liquids, and gases remain intact. In contrast, a molecular equation shows compounds as whole units without breaking them into ions. For example, in a molecular equation, an aqueous compound like sodium chloride (NaCl) is written as NaCl, but in a complete ionic equation, it is written as . This distinction is important because only aqueous compounds dissociate into ions, which is determined using solubility rules. Understanding this difference helps in analyzing the behavior of ions during chemical reactions, especially in precipitation and acid-base reactions.
To write a complete ionic equation from a molecular equation, first identify which compounds are aqueous and will dissociate into ions. Use solubility rules to determine this. Then, break these aqueous compounds into their constituent ions, making sure to distribute coefficients to each ion correctly. For example, if the molecular equation has 2 moles of , write 2 moles of and 2 moles of . Solids, liquids, and gases remain as whole compounds. This process helps reveal all ions present in the reaction, which is essential for further steps like writing net ionic equations.
Spectator ions are ions that appear unchanged on both the reactant and product sides of a complete ionic equation. They do not participate directly in the chemical reaction. When writing a net ionic equation, these spectator ions are removed to show only the ions involved in the actual chemical change. For example, in the reaction between sodium chloride and silver nitrate, and are spectator ions and are omitted in the net ionic equation. This simplification highlights the species that form the precipitate or undergo a chemical transformation.
Understanding the relationship between molecular, complete ionic, and net ionic equations is crucial for analyzing chemical reactions accurately. The molecular equation shows the overall reaction with compounds intact. The complete ionic equation breaks down aqueous compounds into ions, revealing all species present. The net ionic equation further simplifies this by removing spectator ions, focusing only on the ions that participate in the reaction. This stepwise approach helps predict reaction outcomes, especially in acid-base and precipitation reactions, by identifying the active ions responsible for the chemical change.
Solubility rules determine which compounds dissociate into ions in aqueous solutions and which remain as solids, liquids, or gases. When writing complete ionic equations, only aqueous compounds are broken into their constituent ions. For example, soluble salts like sodium chloride dissociate into and , while insoluble compounds like barium sulfate remain as solid units. Applying solubility rules ensures that the complete ionic equation accurately represents the species present in solution, which is essential for identifying spectator ions and writing net ionic equations.
