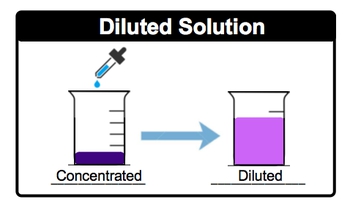
A stock solution is a concentrated solution that can be diluted for various laboratory applications. Dilution involves adding more solvent, typically water, to a solution to decrease its concentration. For instance, when a dark purple solution is gradually mixed with water, the color lightens to a fuchsia hue, indicating a reduction in concentration. This visual change exemplifies the dilution process, where the original solution becomes less concentrated as more solvent is introduced. Understanding this concept is crucial in laboratory settings, as it allows for the preparation of solutions with desired concentrations for experiments and analyses.
- 1. The Chemical World9m
- 2. Measurement and Problem Solving2h 19m
- 3. Matter and Energy2h 15m
- Classification of Matter18m
- States of Matter8m
- Physical & Chemical Changes19m
- Chemical Properties8m
- Physical Properties5m
- Temperature (Simplified)9m
- Law of Conservation of Mass5m
- Nature of Energy5m
- First Law of Thermodynamics7m
- Endothermic & Exothermic Reactions7m
- Heat Capacity17m
- Thermal Equilibrium (Simplified)8m
- Intensive vs. Extensive Properties13m
- 4. Atoms and Elements2h 33m
- The Atom (Simplified)9m
- Subatomic Particles (Simplified)11m
- Isotopes17m
- Ions (Simplified)22m
- Atomic Mass (Simplified)17m
- Periodic Table: Element Symbols6m
- Periodic Table: Classifications11m
- Periodic Table: Group Names8m
- Periodic Table: Representative Elements & Transition Metals7m
- Periodic Table: Phases (Simplified)8m
- Periodic Table: Main Group Element Charges12m
- Atomic Theory9m
- Rutherford Gold Foil Experiment9m
- 5. Molecules and Compounds1h 50m
- Law of Definite Proportions9m
- Periodic Table: Elemental Forms (Simplified)6m
- Naming Monoatomic Cations6m
- Naming Monoatomic Anions5m
- Polyatomic Ions25m
- Naming Ionic Compounds11m
- Writing Formula Units of Ionic Compounds7m
- Naming Acids18m
- Naming Binary Molecular Compounds6m
- Molecular Models4m
- Calculating Molar Mass9m
- 6. Chemical Composition1h 23m
- 7. Chemical Reactions1h 43m
- 8. Quantities in Chemical Reactions1h 8m
- 9. Electrons in Atoms and the Periodic Table2h 32m
- Wavelength and Frequency (Simplified)5m
- Electromagnetic Spectrum (Simplified)11m
- Bohr Model (Simplified)9m
- Emission Spectrum (Simplified)3m
- Electronic Structure4m
- Electronic Structure: Shells5m
- Electronic Structure: Subshells4m
- Electronic Structure: Orbitals11m
- Electronic Structure: Electron Spin3m
- Electronic Structure: Number of Electrons4m
- The Electron Configuration (Simplified)20m
- The Electron Configuration: Condensed4m
- Ions and the Octet Rule9m
- Valence Electrons of Elements (Simplified)5m
- Periodic Trend: Metallic Character4m
- Periodic Trend: Atomic Radius (Simplified)7m
- Periodic Trend: Ionization Energy (Simplified)9m
- Periodic Trend: Electron Affinity (Simplified)7m
- Electron Arrangements5m
- The Electron Configuration: Exceptions (Simplified)12m
- 10. Chemical Bonding2h 10m
- Lewis Dot Symbols (Simplified)7m
- Ionic Bonding6m
- Covalent Bonds6m
- Lewis Dot Structures: Neutral Compounds (Simplified)8m
- Bonding Preferences6m
- Multiple Bonds4m
- Lewis Dot Structures: Multiple Bonds10m
- Lewis Dot Structures: Ions (Simplified)8m
- Lewis Dot Structures: Exceptions (Simplified)12m
- Resonance Structures (Simplified)5m
- Valence Shell Electron Pair Repulsion Theory (Simplified)4m
- Electron Geometry (Simplified)7m
- Molecular Geometry (Simplified)9m
- Bond Angles (Simplified)11m
- Dipole Moment (Simplified)14m
- Molecular Polarity (Simplified)7m
- 11 Gases2h 7m
- 12. Liquids, Solids, and Intermolecular Forces1h 11m
- 13. Solutions3h 1m
- 14. Acids and Bases2h 14m
- 15. Chemical Equilibrium1h 27m
- 16. Oxidation and Reduction1h 33m
- 17. Radioactivity and Nuclear Chemistry53m
Dilutions: Videos & Practice Problems
Dilutions describe making a solution less concentrated by adding more solvent, usually water. A concentrated stock solution or standard is diluted to produce a lower molarity. In this process, the amount of solute stays the same while the concentration decreases, so the initial concentration is greater than the final concentration: \(M_1V_1=M_2V_2\) .
Here, \(M_1\) and \(V_1\) are the molarity and volume before dilution, and \(M_2\) and \(V_2\) are after dilution. Because solvent is added, \(M_1\) is always larger than \(M_2\), and the final volume is found from \(V_2=V_1+\text{volume of solvent added}\) . A common sign of a dilution problem is working with one compound at two different molarities. Some situations involve a series of dilutions, where the same relationship is applied step by step to track the concentration after each dilution.
In Dilutions, a solvent (usually water) is added to a concentrated solution.
Concentrated & Diluted Solutions
Dilutions
Dilutions Video Summary

Dilutions Example 1
Dilutions Example 1 Video Summary
To determine the concentration of solutions based on the number of solute moles represented by spheres, we can calculate the molarity for each solution. Molarity (M) is defined as the number of moles of solute divided by the volume of solution in liters:
Molarity (M) = \(\frac{\text{moles of solute}}{\text{liters of solution}}\)
In this example, we have three solutions:
Solution A: Contains 5 spheres (moles of solute) in 1 liter of solution. Thus, the molarity is:
M_A = \(\frac{5 \text{ moles}}{1 \text{ L}} = 5 \text{ M}\)
Solution B: Contains 3 spheres in 2 liters of solution. Therefore, the molarity is:
M_B = \(\frac{3 \text{ moles}}{2 \text{ L}} = 1.5 \text{ M}\)
Solution C: Contains 6 spheres in 3 liters of solution, giving us:
M_C = \(\frac{6 \text{ moles}}{3 \text{ L}} = 2 \text{ M}\)
Now, to arrange the solutions from least concentrated to most concentrated based on their molarity values, we find:
1. Solution B: 1.5 M
2. Solution C: 2 M
3. Solution A: 5 M
Thus, the order from least concentrated to most concentrated is B, C, and A.
Dilutions
Dilutions Video Summary
Understanding dilution is essential in chemistry, as it allows us to create solutions with lower concentrations from more concentrated ones. The process of dilution can be quantitatively described using the equation:
\( M_1 V_1 = M_2 V_2 \)
In this equation, \( M_1 \) and \( V_1 \) represent the molarity and volume of the solution before dilution, while \( M_2 \) and \( V_2 \) represent the molarity and volume after dilution. It is important to note that \( M_1 \), the molarity of the concentrated solution, is always greater than \( M_2 \), the molarity of the diluted solution.
The final volume after dilution, \( V_2 \), is determined by the initial volume \( V_1 \) plus the volume of solvent added. This relationship can be expressed as:
\( V_2 = V_1 + V_{\text{solvent}} \)
By applying these principles, one can effectively prepare solutions with desired concentrations, which is a fundamental skill in various scientific applications.

Dilutions Example 2
Dilutions Example 2 Video Summary
To determine the volume of a concentrated solution needed to prepare a diluted solution, we can apply the dilution equation, which is expressed as:
\( M_1 V_1 = M_2 V_2 \)
In this equation, \( M_1 \) represents the molarity of the concentrated solution, \( V_1 \) is the volume of the concentrated solution we need to find, \( M_2 \) is the molarity of the diluted solution, and \( V_2 \) is the volume of the diluted solution.
In the given problem, we have:
- Concentrated solution: 5.2 M (this is \( M_1 \))
- Diluted solution: 2.7 M (this is \( M_2 \))
- Volume of diluted solution: 3.5 L (this is \( V_2 \))
Since we are dealing with one compound, hydrobromic acid, and two different molarities, this indicates a dilution scenario. To find \( V_1 \), we rearrange the equation:
\( V_1 = \frac{M_2 V_2}{M_1} \)
Substituting the known values into the equation gives:
\( V_1 = \frac{(2.7 \, \text{M})(3.5 \, \text{L})}{5.2 \, \text{M}} \)
Calculating this yields:
\( V_1 = \frac{9.45 \, \text{mol}}{5.2 \, \text{M}} = 1.8173 \, \text{L} \)
To convert liters to milliliters, we use the conversion factor where 1 L = 1000 mL:
\( 1.8173 \, \text{L} \times 1000 \, \text{mL/L} = 1817.3 \, \text{mL} \)
Considering significant figures, since the values 5.2, 3.5, and 2.7 all have two significant figures, we round 1817.3 mL to 1800 mL. Thus, the final answer is:
1800 mL
In summary, when faced with a dilution problem involving a single compound and two molarities, the dilution equation is the key to finding the unknown volume of the concentrated solution needed for preparation.
To what final volume would 100 mL of 5.0 M KCl have to be diluted in order to make a solution that is 0.54 M KCl?
If 880 mL of water is added to 125.0 mL of a 0.770 M HBrO4 solution what is the resulting molarity?
A student prepared a stock solution by dissolving 25.00 g of NaOH in enough water to make 150.0 mL solution. The student took 20.0 mL of the stock solution and diluted it with enough water to make 250.0 mL solution. Finally taking 75.0 mL of that solution and dissolving it in water to make 500 mL solution. What is the concentration of NaOH for this final solution? (MW of NaOH:40.00 g/mol).
Do you want more practice?
More setsGo over this topic definitions with flashcards
More setsHere's what students ask on this topic:
A stock solution is a concentrated solution that serves as the starting point for preparing diluted solutions. It is important because it allows chemists to create solutions of various concentrations by adding solvent, usually water, to the stock. This process is called dilution. Using a stock solution is efficient and practical in the laboratory since it avoids the need to prepare multiple solutions from scratch. Instead, a single concentrated stock can be diluted to the desired concentration for different experiments or applications.
Dilution decreases the concentration of a solution by adding more solvent, typically water, to the original solution. This increases the total volume but keeps the amount of solute constant, resulting in a lower molarity. For example, if you start with a dark purple concentrated solution and add water, the color becomes lighter, indicating a decrease in concentration. The relationship between the initial and final concentrations and volumes is given by the equation , where and are the molarity and volume before dilution, and and are after dilution.
The equation relates the molarity and volume before and after dilution. To use it, you need to know three of the four variables: initial molarity (), initial volume (), final molarity (), or final volume (). For example, if you know the initial concentration and volume of a stock solution and the desired final concentration, you can calculate the volume of the diluted solution needed. Remember, is the total volume after adding solvent, so . This equation helps in planning how much solvent to add to achieve the desired dilution.
The molarity of the stock solution () is always greater than the molarity after dilution () because dilution involves adding solvent to the original solution. Adding solvent increases the total volume but does not change the amount of solute present. Since molarity is defined as moles of solute per liter of solution, increasing the volume while keeping moles constant decreases the molarity. This is why the concentration decreases, and the solution becomes less concentrated after dilution.
To calculate the volume of solvent to add during dilution, you first use the dilution equation to find the final volume . Once you have , subtract the initial volume from it to get the volume of solvent to add: . This calculation ensures you add the correct amount of solvent to achieve the desired lower concentration.
