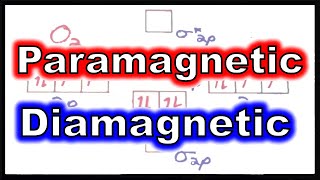
10. Periodic Properties of the Elements
Paramagnetism and Diamagnetism
10. Periodic Properties of the Elements
Paramagnetism and Diamagnetism
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
Which of the following atoms has the most unpaired electrons?
5views1rank - Multiple Choice
Write the condensed electron configuration for the nickel (III) ion and state if it is paramagnetic or diamagnetic.
2views1rank - Multiple Choice
Write the condensed electron configuration for the copper (I) ion and is it magnetic?
4views1rank - Multiple ChoiceWhich one of the following atoms or ions is diamagnetic?4views
- Open Question
In the ground-state electron configuration of Fe3+, how many unpaired electrons are present?
1views - Open QuestionSelect the element(s) that will have one unpaired electron in the p orbital.
- Open QuestionIdentify the general outer electron configuration for each group of elements shown in this periodic table outline.1views
- Open QuestionSort the following atom or ions as paramagnetic or diamagnetic according to the electron configurations determined in part a.1views