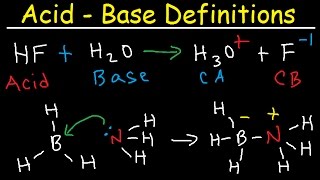
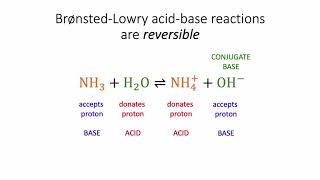17. Acid and Base Equilibrium
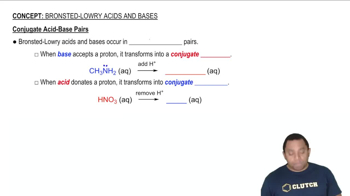
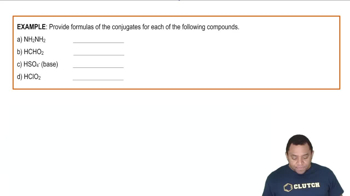
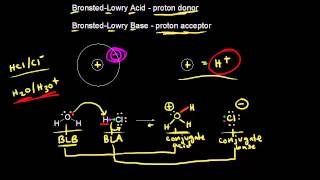
Bronsted-Lowry Acids and Bases
17. Acid and Base Equilibrium
Bronsted-Lowry Acids and Bases
Showing 9 of 9 videos
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
Write the formula of the conjugate base for the following compound:
H2Se
10views - Multiple Choice
Write the formula of the conjugate for the following compound:
NH2NH2
4views - Multiple Choice
Which of the following is a Bronsted-Lowry acid?
5views1rank - Multiple Choice
Determine the chemical equation that would result when carbonate, CO32-, reacts with water.
4views1rank - Open Question
In the reversible reaction HCN + H2O ⇌ CN- + H3O+, the two Bronsted-Lowry acids are...
1views - Open QuestionH2co3(aq)+h2o(l)⇌h3o+(aq)+hco3−(aq)1views
- Open Question
Write the formula of the conjugate acid of the Brønsted-Lowry base, (CH3)3N
1views - Open QuestionWrite the formula for the conjugate acid of each of the following bases.1views