9. Quantum Mechanics
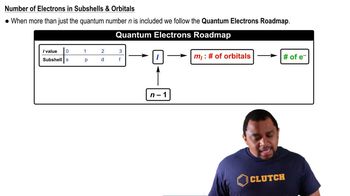
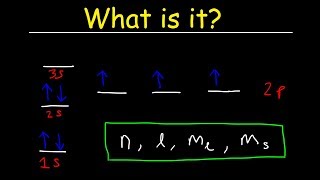
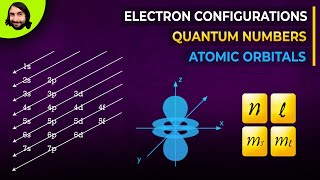
Quantum Numbers: Number of Electrons
9. Quantum Mechanics
Quantum Numbers: Number of Electrons
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
Determine the number of electrons that can have the following set of quantum numbers:n = 3, ml = 0.
2views - Multiple Choice
Determine the number of electrons that can have the following set of quantum numbers:n = 2, ms = –1/2.
2views - Multiple Choice
Determine the number of electrons that can have the following set of quantum numbers.
n = 4, l = 3, ml = – 1
1views - Multiple Choice
Determine the number of electrons that can have the following set of quantum numbers.
n = 4, mL = – 1, ms = –1/2
1views - Open QuestionWhat is the maximum number of electrons that can occupy each of the following subshells?2views
- Open Question
What is the maximum number of electrons that can exist in the g sublevel?
- Open Question
Enter the maximum number of electrons in each type of sublevel (s, p, d, and f, respectively).
1views - Open Question
What's the maximum amount of electrons that will fit on an outermost shell of an atom?
1views