Which of the following pairs of compounds could be separated by recrystallization or distillation?
a. meso-tartaric acid and (±)-tartaric acid (HOOC—CHOH—CHOH—COOH)

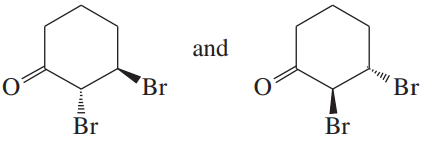
 Verified step by step guidance
Verified step by step guidance
Which of the following pairs of compounds could be separated by recrystallization or distillation?
a. meso-tartaric acid and (±)-tartaric acid (HOOC—CHOH—CHOH—COOH)
Which of the following pairs of compounds could be separated by recrystallization or distillation?
(b)
For each structure,
1. star (*) any asymmetric carbon atoms.
2. label each asymmetric carbon as (R) or (S).
3. draw any internal mirror planes of symmetry.
4. label the structure as chiral or achiral.
5. label any meso structures.
(a)
(b)
The following four structures are naturally occurring optically active compounds. Star (*) the asymmetric carbon atoms in these structures.
To show that (R)-2-butyl (R,R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.