
Draw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you.
(i) 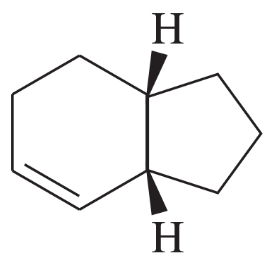
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Asymmetric Carbon Atoms
Enantiomers

Molecular Models
Make a model and draw a three-dimensional structure for each compound. Then draw the mirror image of your original structure and determine whether the mirror image is the same compound. Label each structure as being chiral or achiral, and label pairs of enantiomers.
(e)
(f)
For each of the stereocenters (circled) in Figure 5-5,
a. draw the compound with two of the groups on the stereocenter interchanged.
b. give the relationship of the new compound to the original compound.
Draw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you.
(c)
(d) 1-bromo-2-methylbutane
Draw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you
(a)
(b)
