Textbook Question
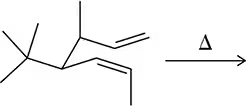
Predict the product of the Claisen reactions shown.
(b)
2
views

 Verified step by step guidance
Verified step by step guidance

Predict the product of the Claisen reactions shown.
(b)
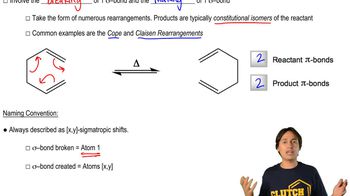
Predict the product of the following reactions. [When all of the reactions from this chapter are shown together, you must first decide which type of reaction each is. Is it a Diels–Alder, an electrocyclic, or a sigmatropic rearrangement? Drawing the product will be easier once this determination is made.]
(a)
Draw the products you’d expect to form in Assessment 22.38.
(c)
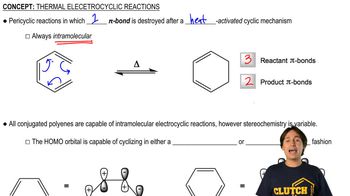
Given the conditions, would you expect conrotatory or disrotatory ring closing/opening? Justify this on the basis of the molecular orbital picture.
(c)
Predict the product of the Cope reactions shown.
(a)
Predict the product of the Claisen reactions shown.
(c)