Textbook Question
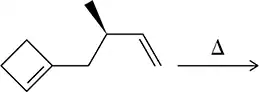
Predict the product of the Claisen reactions shown.
(b)
2
views

 Verified step by step guidance
Verified step by step guidance

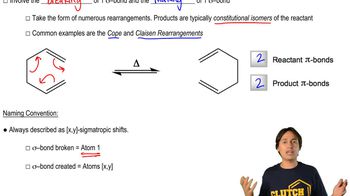
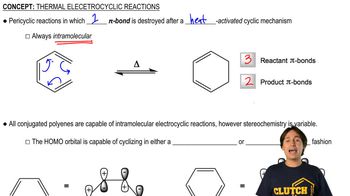
Predict the product of the Claisen reactions shown.
(b)
Draw the products you’d expect to form in Assessment 22.38.
(c)
Given the conditions, would you expect conrotatory or disrotatory ring closing/opening? Justify this on the basis of the molecular orbital picture.
(c)
Predict the product of the Claisen reactions shown.
(c)
For the following electrocyclic reactions, did the substituents move in a conrotatory or disrotatory direction? Would you use heat or light to cause movement in this direction?
(a)
Predict the product of the Cope reactions shown.
(c)