Textbook Question
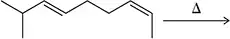
Predict the product of the following electrocyclic reactions.
(a)
2
views

 Verified step by step guidance
Verified step by step guidance
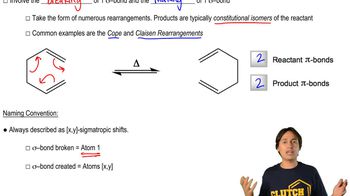
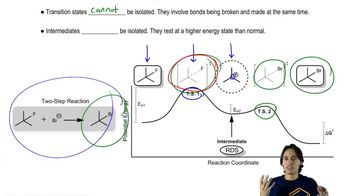

Predict the product of the following electrocyclic reactions.
(a)
Identify the following dienes as being in the s-cis or s-trans conformation. If they are in the s-trans conformation, draw them in the s-cis conformation. [It may not always be possible.]
(e)
Predict the product of the following electrocyclic reactions.
(c)
Predict the product of the following sigmatropic rearrangements. Be sure to rationalize the stereochemical outcome with a chair-like transition state.
(d)
Identify the following dienes as being in the s-cis or s-trans conformation. If they are in the s-trans conformation, draw them in the s-cis conformation. [It may not always be possible.]
(c)
Predict the product of the following electrocyclic reactions.
(b)