Textbook Question
Identify each of the following reactions as an oxidation reaction, a reduction reaction, or neither.
a.
b.
10
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 27d
Problem 27d Verified step by step guidance
Verified step by step guidance

Identify each of the following reactions as an oxidation reaction, a reduction reaction, or neither.
a.
b.
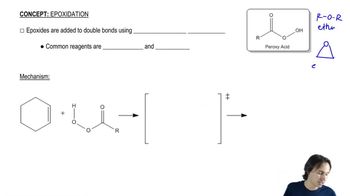
What alkene would you treat with a peroxyacid in order to obtain each of the epoxides in Problem 27?
c. 2,2,3,3-tetramethyloxirane
d. 2,3-epoxy-2-methylpentane
Draw structures for the following:
a. 2-propyloxirane
b. cyclohexene oxide
What alkene would you treat with a peroxyacid in order to obtain each of the epoxides in Problem 27?
a. 2-propyloxirane
b. cyclohexene oxide
What will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carried out in
c. ethyl alcohol?
d. methyl alcohol?
Draw structures for the following:
c. 2,2,3,3-tetramethyloxirane