 Back
BackIntroduction to Biochemistry: Foundations, Molecules, and Cellular Organization
Study Guide - Smart Notes

Introduction to Biochemistry
What is Biochemistry?
Biochemistry is the scientific discipline that explores the chemical processes and substances that occur within living organisms. It seeks to answer fundamental questions such as “What are we made of?” and “How do we work?” by examining both the structures and the physical and chemical processes that sustain life.
Physical processes: Changes that do not alter the chemical composition of a substance (e.g., melting or evaporating water).
Chemical processes: Changes that result in a new chemical composition (e.g., electrolysis of water).
Biochemistry is inherently multidisciplinary, integrating biology, chemistry, physics, and more.
Applications include medicine, industry, agriculture, technology, and nutrition.


Characteristics of Life
Defining Life and Its Organization
All living organisms share a set of characteristics that distinguish them from nonliving matter. These include organization, metabolism, homeostasis, growth, response to stimuli, reproduction, and evolution.
Cellular organization: Life is organized in a hierarchy from atoms to molecules, organelles, cells, tissues, organs, organ systems, and organisms.
Biochemical unity: All living organisms are remarkably similar at the molecular and atomic levels, supporting the theory of a common universal ancestor.
Viruses: Not considered alive because they lack several characteristics of life, such as cellular structure and independent metabolism.

Abiogenesis: The Origin of Life
How Life Began from Nonliving Matter
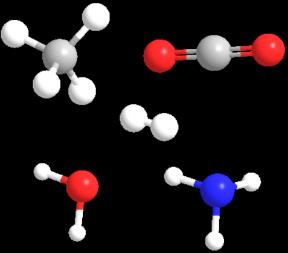
Abiogenesis describes the natural process by which life arose from nonliving, simple molecules. Life is thought to have originated in Earth’s oceans about 3.8 billion years ago from prebiotic materials such as H2, H2O, NH3, CO2, and CH4.
Atmospheric conversion theory: Lightning and solar radiation converted prebiotics to simple biomolecules.
Hydrothermal vent theory: Vents on the ocean floor provided energy for the conversion of prebiotics to biomolecule monomers.
Polymerization on mineral surfaces: Charged mineral surfaces (e.g., clay) facilitated the alignment and polymerization of biomolecules.
Membrane formation: Lipid membranes enclosed pools of biomolecules, forming protocells and increasing the likelihood of molecular interactions.
Double Origin Theory: Coding systems and enzyme-catalysis developed in separate protocells and later combined.
RNA World Hypothesis: RNA was likely the first genetic material due to its ability to encode information and catalyze reactions.


Nucleic Acids
Structure and Function of Nucleic Acids
Nucleic acids are one of the four major biological macromolecules and are essential for storing and transmitting hereditary information. They are polymers of nucleotide monomers, each consisting of a phosphate group, a pentose sugar, and a nitrogenous base.
DNA (deoxyribonucleic acid) and RNA (ribonucleic acid) are the primary nucleic acids in cells.
Nucleic acids have directionality, with a 5’ end and a 3’ end.
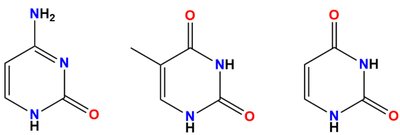
Nitrogenous bases are classified as purines (adenine, guanine) or pyrimidines (cytosine, thymine, uracil).
Base-pairing occurs via hydrogen bonds according to Watson-Crick rules (A-T/U, G-C).
Proteins
Structure and Function of Proteins
Proteins are versatile macromolecules that perform a wide range of structural and functional roles in living organisms. They are polymers of amino acids, each with a central carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique R-group.
Proteins have directionality: the N-terminal (amino group) and C-terminal (carboxyl group).
There are 20 standard amino acids, classified by their R-groups.
Protein structure is organized into four levels: primary (sequence), secondary (α-helices, β-sheets), tertiary (3D shape), and quaternary (multiple polypeptide chains).
Enzymes are proteins that catalyze biochemical reactions without being consumed.
Carbohydrates
Structure and Function of Carbohydrates
Carbohydrates are sugars composed mainly of carbon, hydrogen, and oxygen, typically with the formula Cn(H2O)n. They serve as primary short-term energy sources and structural components in cells.
Monosaccharides: Simple sugar monomers (e.g., glucose).
Polysaccharides: Polymers of monosaccharides (e.g., starch, glycogen, cellulose, chitin, peptidoglycan).
Carbohydrates can be linked to proteins (glycoproteins), lipids (glycolipids), and nucleic acids (nucleotides).
Lipids
Structure and Function of Lipids
Lipids are a diverse group of hydrophobic molecules, including fats, oils, waxes, phospholipids, and steroids. They are important for energy storage, membrane structure, and signaling.
Phospholipids are amphipathic, containing both hydrophilic (polar head) and hydrophobic (nonpolar tail) regions, enabling membrane formation.
Cell membranes are composed of a phospholipid bilayer with embedded proteins and other molecules.
Only small, nonpolar molecules can freely cross the membrane without energy input.
Taxonomy and Domains of Life
Classification of Living Organisms
Taxonomy is the science of classifying, identifying, and naming living organisms. Life is organized into hierarchical categories: Domain, Kingdom, Phylum, Class, Order, Family, Genus, Species.
Three domains: Bacteria, Archaea (both prokaryotic), and Eukarya (eukaryotic).
Prokaryotes lack a membrane-bound nucleus; eukaryotes have one.
Cell Organelles
Structure and Function of Organelles
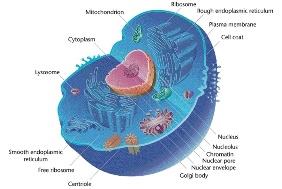
Cells contain specialized subcellular structures called organelles. Eukaryotic cells have membrane-bound organelles, while prokaryotic cells do not.
Key organelles: nucleus, mitochondria, chloroplasts, endoplasmic reticulum, Golgi apparatus, lysosomes, ribosomes.
Ribosomes are involved in protein synthesis (translation) and differ in size between prokaryotes (70S) and eukaryotes (80S).

Endosymbiotic Theory
Origin of Mitochondria and Chloroplasts
The endosymbiotic theory proposes that mitochondria and chloroplasts originated as free-living bacteria that were engulfed by ancestral eukaryotic cells. Over time, these bacteria became integrated as organelles.
Evidence: Both organelles have circular DNA, 70S ribosomes, and replicate by binary fission.
Mitochondria are the site of aerobic respiration; chloroplasts perform photosynthesis.

Central Dogma of Molecular Biology
Flow of Genetic Information
The central dogma describes the flow of genetic information from DNA to RNA (transcription) and from RNA to protein (translation). This process is unidirectional and fundamental to all living organisms.
Transcription: DNA is used as a template to synthesize RNA.
Translation: mRNA is decoded by ribosomes to build proteins.
Functional Groups in Biomolecules
Common Functional Groups and Linkages
Functional groups are specific groups of atoms within molecules that are responsible for characteristic chemical reactions. They are abundant in biomolecules and include amino, carboxyl, hydroxyl, phosphate, and methyl groups.
Proteins: amino and carboxyl groups
Carbohydrates: hydroxyl groups and ether linkages
Lipids: carboxyl groups (in fatty acids)
Nucleic acids: phosphodiester linkages
Chemical Bonds
Types of Bonds in Biochemistry
Chemical bonds are the forces that hold atoms together in molecules. They are classified as intramolecular (within molecules) or intermolecular (between molecules).
Ionic bonds: Attraction between oppositely charged ions.
Covalent bonds: Sharing of electron pairs between atoms (can be polar or nonpolar).
Hydrogen bonds: Weak interactions involving hydrogen and electronegative atoms (N, O, F).
Van der Waals forces: Weak attractions due to temporary dipoles.
Organic Chemistry in Biochemistry
Carbon Compounds and Stereochemistry
Organic chemistry focuses on carbon-containing compounds, which are central to biochemistry. Stereochemistry examines the spatial arrangement of atoms, including isomers, chiral centers, and enantiomers.
Isomers: Molecules with the same formula but different structures.
Chirality: Carbon atoms bonded to four different groups, leading to non-superimposable mirror images (enantiomers).
Resonance: Delocalization of electrons within a molecule, stabilizing its structure.
Thermodynamics in Biochemistry
Entropy, Free Energy, and Equilibrium
Thermodynamics governs the energy changes in biochemical reactions. Entropy measures disorder, while Gibbs free energy determines the spontaneity of reactions.
Gibbs Free Energy Equation:
Spontaneous reactions: Exergonic (negative ΔG), increase universal entropy.
Nonspontaneous reactions: Endergonic (positive ΔG), require energy input.
Equilibrium constant (Keq): Ratio of product to reactant concentrations at equilibrium.
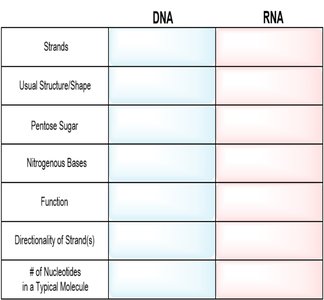
Summary Table: Comparison of DNA and RNA
Feature | DNA | RNA |
|---|---|---|
Strands | Usually double-stranded | Usually single-stranded |
Usual Structure/Shape | Double helix | Varies (often single helix) |
Pentose Sugar | Deoxyribose | Ribose |
Nitrogenous Bases | A, T, C, G | A, U, C, G |
Function | Stores hereditary information | Encodes proteins, catalytic roles |
Directionality | 5’ → 3’ | 5’ → 3’ |
# of Nucleotides | Hundreds to thousands | Varies (shorter) |
