Textbook Question
Suggest a synthesis of the following acylated sugars.
(b)

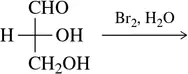
 Verified step by step guidance
Verified step by step guidance
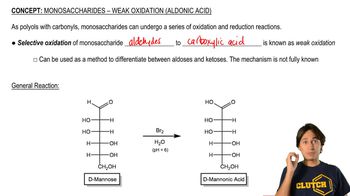
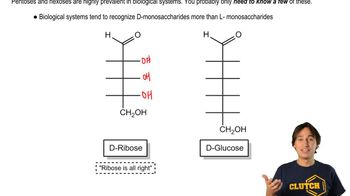
Suggest a synthesis of the following acylated sugars.
(b)
Suggest a synthesis of the following acylated sugars.
(a)
Predict the product of each of the following reactions.
(b)
In each of the disaccharides shown (a–c), (i) identify the reducing end of the sugar, (ii) tell what monosaccharides are used to make it, and (iii) tell what type of glycoside linkage is present.
(a)
Ketohexoses form from aldohexoses. Suggest a mechanism for the acid-catalyzed version of this reaction. What type of reaction is this? [You’ve seen it before.]
Predict the product of each of the following reactions.
(d)