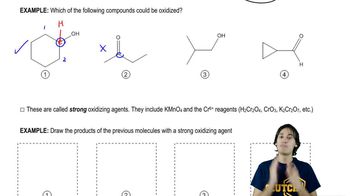
Except for the Tollens test, basic aqueous conditions are generally avoided with sugars because they lead to fast isomerizations.
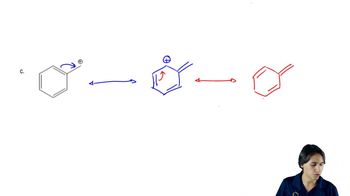
(a) Under basic conditions, the proton alpha to the aldehyde (or ketone) carbonyl group is reversibly removed, and the resulting enolate ion is no longer asymmetric. Reprotonation can occur on either face of the enolate, giving either the original structure or its epimer. Because a mixture of epimers results, this process is called epimerization. Propose a mechanism for the base-catalyzed equilibration of glucose to a mixture of glucose and its C2 epimer, mannose.