Textbook Question
In the following reactions, identify the bonds formed and the bonds broken.
(c)
2
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 9
Problem 9 Verified step by step guidance
Verified step by step guidance
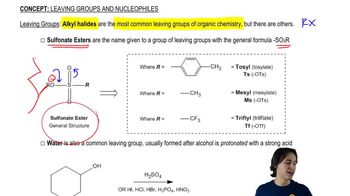
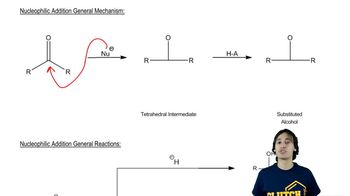

In the following reactions, identify the bonds formed and the bonds broken.
(c)
Practice your electron-pushing skills by drawing a mechanism for the following SN2 reactions.
(b)
For the following molecule, draw the Newman projection (around the 2,3-bond) with a dihedral angle of 180° between the bromine and hydrogen.
Given the following rate data, how many molecules are in the rate-determining step of this reaction?
In the following reactions, identify the bonds formed and the bonds broken.
(a)
Practice your electron-pushing skills by drawing a mechanism for the following SN2 reactions.
(a)