Show how you would synthesize the following ethers in good yield from the indicated starting materials and any additional reagents needed.
(a) cyclopentyl n-propyl ether from cyclopentanol and propan-1-ol

 Verified step by step guidance
Verified step by step guidance

Show how you would synthesize the following ethers in good yield from the indicated starting materials and any additional reagents needed.
(a) cyclopentyl n-propyl ether from cyclopentanol and propan-1-ol
Show how you would synthesize the following ethers in good yield from the indicated starting materials and any additional reagents needed.
(c) 2-ethoxyoctane from an octene
Show how you would synthesize the following ethers in good yield from the indicated starting materials and any additional reagents needed.
(b) n-butyl phenyl ether from phenol and butan-1-ol
Give the structures of intermediates A through H in the following synthesis of trans-1-cyclohexyl-2-methoxycyclohexane.
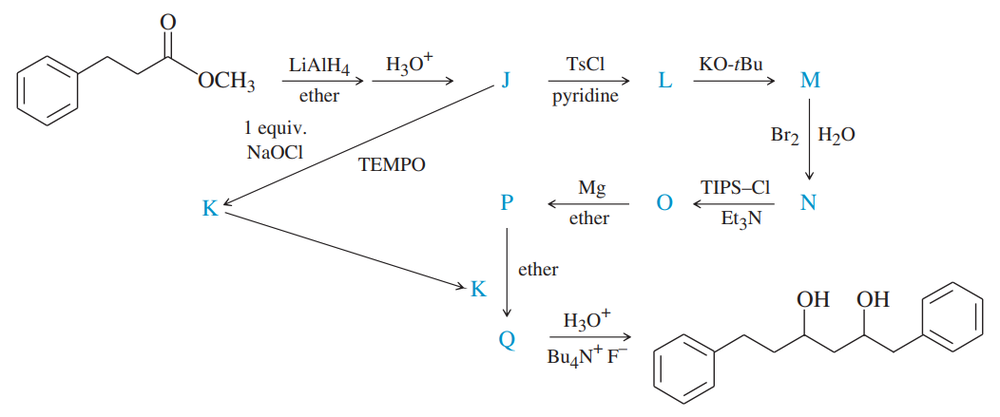
Give the structures of the intermediates represented by letters K and Q in this synthesis.
Give the structures of the intermediates represented by letters J, L, M, and N in this synthesis.