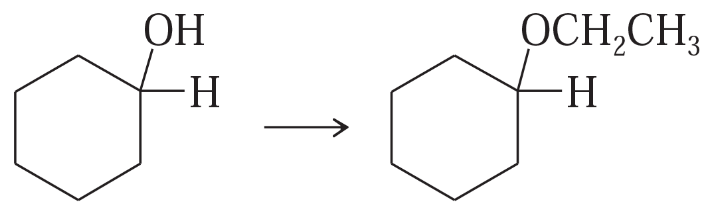
Show how you would accomplish the following synthetic conversions.
(d)

 Verified step by step guidance
Verified step by step guidance


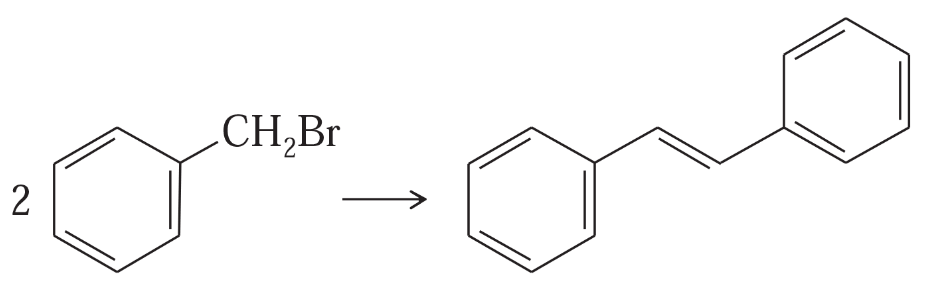
Show how you would accomplish the following synthetic conversions.
(d)
In each case, show how you would synthesize the chloride, bromide, and iodide from the corresponding alcohol.
(a) 1-halobutane (halo = chloro, bromo, iodo)
(b) halocyclopentane
Show how you would convert 2-methylcyclopentanol to the following products. Any of these products may be used as the reactant in any subsequent part of this problem.
(g) 2-methylcyclopentyl acetate
(h) 1-bromo-1-methylcyclopentane
Show how you would convert 2-methylcyclopentanol to the following products. Any of these products may be used as the reactant in any subsequent part of this problem.
d. 1-methylcyclopentanol
Predict the major products of dehydration catalyzed by sulfuric acid.
(a) hexan-1-ol
(b) hexan-2-ol
(c) pentan-3-ol
Show how you would accomplish the following synthetic conversions.
(c)