Textbook Question
Determine the degree of unsaturation and then draw possible structures for noncyclic compounds with the following molecular formulas:
a. C3H6
4
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics
Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics Problem 5b
Problem 5b Verified step by step guidance
Verified step by step guidance
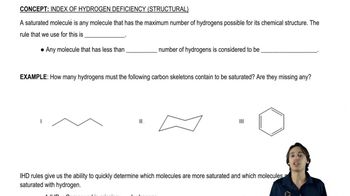


Determine the degree of unsaturation and then draw possible structures for noncyclic compounds with the following molecular formulas:
a. C3H6
What is each compound's systematic name?
c.
d.
Several studies have shown that β-carotene, a precursor of vitamin A, may play a role in preventing cancer. β-Carotene has a molecular formula of C40H56, and it contains two rings and no triple bonds. How many double bonds does it have?
What is the molecular formula for each of the following?
b. a 10-carbon hydrocarbon with one bond and 2 rings
What is each compound's systematic name?
a.
b.
Determine the degree of unsaturation for hydrocarbons with the following molecular formulas:
c. C12H20
d. C40H56