For each of the following ketones/aldehydes, indicate whether it is possible to synthesize it from an alkyne as the only compound in good ( > 50%) yield. If so, how would you do it?
(a)

 Mullins 1st Edition
Mullins 1st Edition Ch. 10 - Alkynes: Electrophilic Addition and Redox Reactions
Ch. 10 - Alkynes: Electrophilic Addition and Redox Reactions Problem 37
Problem 37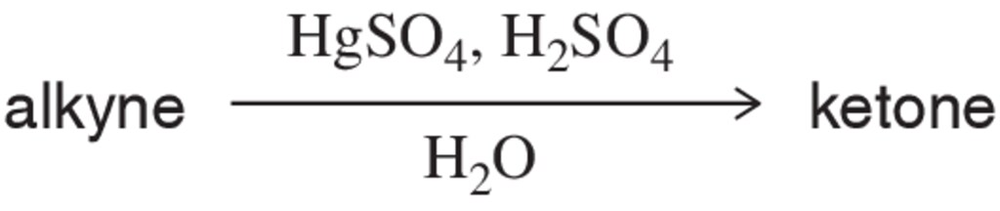
 Verified step by step guidance
Verified step by step guidance
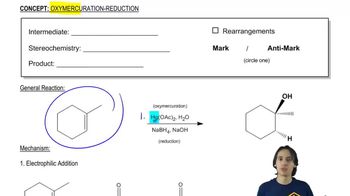

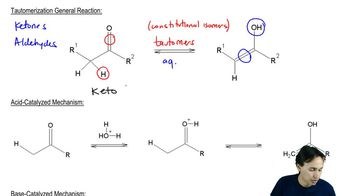
For each of the following ketones/aldehydes, indicate whether it is possible to synthesize it from an alkyne as the only compound in good ( > 50%) yield. If so, how would you do it?
(a)
For each of the following ketones/aldehydes, indicate whether it is possible to synthesize it from an alkyne as the only compound in good (> 50%) yield. If so, how would you do it?
(d)
Predict the major products resulting from the addition of one equivalent of HX to the following alkynes.
(b)
Predict the alkyne and reactants you might use to make the following haloalkenes. [Providing the reactant and the reagent is how we start thinking about synthesis.]
(a)
Predict the alkyne and reactants you might use to make the following haloalkenes. [Providing the reactant and the reagent is how we start thinking about synthesis.]
(b)
Draw the ketone(s) you would expect to form by treating the following alkynes under the conditions of hydroboration–oxidation: (a) 6-methyloct-1-yne, (b) 1,10-dicyclohexyldec-5-yne, and (c) 5-phenylhex-2-yne.
<IMAGE>