Textbook Question
Which of the carbocations in part a is most stable?
1. the isobutyl cation?
2. the n-butyl cation?
3. the sec-butyl cation?
3
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 2b
Problem 2b Verified step by step guidance
Verified step by step guidance
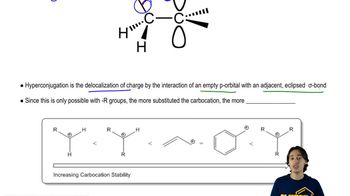
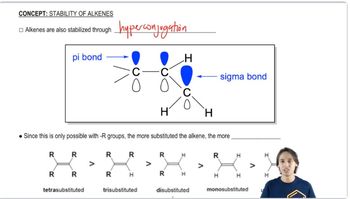

Which of the carbocations in part a is most stable?
1. the isobutyl cation?
2. the n-butyl cation?
3. the sec-butyl cation?
Draw the mechanism for the reaction of cyclohexene with HCl
How many s bond orbitals are available for overlap with the vacant p orbital in
1. the isobutyl cation?
2. the n-butyl cation?
3. the sec-butyl cation?
Rank the following carbocations in each set from most stable to least stable:
a.
How many s bond orbitals are available for overlap with the vacant p orbital in the methyl cation?