 Back
BackMatter and Measurements: Foundations of General Chemistry
Study Guide - Smart Notes

Matter and Measurements
Classification of Matter
Chemistry is the study of matter and the changes it undergoes. The atom is the basic functional unit of matter. Matter is anything that occupies space and has mass. It can be classified into three main types:
Element: The simplest type of matter, composed of one kind of atom.
Compound: Matter composed of two or more different elements that are chemically bonded together.
Mixture: Matter composed of elements and/or compounds that are physically mixed together, not chemically bonded.
Mixtures can be further classified as homogeneous (uniform composition throughout) or heterogeneous (composition varies throughout).
Physical and Chemical Changes
Changes in matter can be classified as physical or chemical:
Physical Change: A change in the physical state or appearance of a substance without altering its composition (e.g., melting, freezing, dissolving).
Chemical Change: A change that results in the formation of new chemical substances with different properties (e.g., burning, rusting, reacting with acids).
Physical changes are often reversible, while chemical changes are usually irreversible.
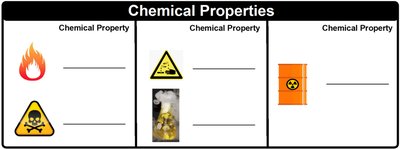
Chemical Properties
Chemical properties describe how a substance interacts with other substances, resulting in a change in composition. Examples include flammability, reactivity with acids, and radioactivity.
Physical Properties
Physical properties are measurable characteristics that describe the state of a substance without changing its chemical structure. Examples include color, mass, density, melting point, and boiling point.
Color: The visual appearance of a substance.
Mass: The amount of matter in an object, measured in kilograms (kg).
Shape: The geometric form of a substance (e.g., cube, diamond).


Intensive vs. Extensive Properties
Properties of matter can be classified as intensive or extensive:
Intensive Properties: Independent of the amount of substance present (e.g., color, density, melting point, luster).
Extensive Properties: Dependent on the amount of substance present (e.g., mass, volume, length).


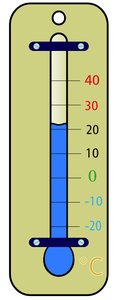
Temperature and Heat
Temperature is a measure of the average kinetic energy of particles in a substance, while heat is the transfer of thermal energy from a hotter object to a cooler one. Temperature is measured in degrees Celsius (ºC), Fahrenheit (ºF), or Kelvin (K).

Scientific Notation
Scientific notation is used to express very large or very small numbers in a compact form:
Format: $a \times 10^n$ where $1 \leq a < 10$ and $n$ is an integer.
Example: $6.02 \times 10^{23}$
SI Units and Metric Prefixes
The International System of Units (SI) is based on seven base units, including kilogram (kg) for mass, meter (m) for length, second (s) for time, and kelvin (K) for temperature. Metric prefixes indicate multiples or fractions of base units (e.g., kilo-, centi-, milli-).


Significant Figures
Significant figures reflect the precision of a measured value. The rules for counting significant figures include:
All nonzero digits are significant.
Zeros between significant digits are significant.
Leading zeros are not significant.
Trailing zeros in a decimal number are significant.
Density
Density is the amount of mass per unit volume. It is calculated as:
$\text{Density} = \frac{\text{Mass}}{\text{Volume}}$
For solids and liquids: units are typically g/cm3 or kg/L.
For gases: units are typically g/L.
Water Displacement
Water displacement is a method used to determine the volume of irregularly shaped objects by measuring the volume of water displaced when the object is submerged.

Dimensional Analysis
Dimensional analysis is a systematic approach to problem-solving that uses conversion factors to move from one unit to another. The process involves:
Identifying the given amount and the desired unit.
Writing down all relevant conversion factors.
Multiplying through by conversion factors to cancel unwanted units.
Practice and Application
Throughout the study of matter and measurements, practice problems reinforce the concepts of classification, physical and chemical changes, properties, significant figures, and unit conversions. Mastery of these foundational topics is essential for success in general chemistry and related laboratory work.
