 Back
BackGeneral Chemistry Study Guide: Key Concepts and Practice Problems
Study Guide - Smart Notes

Q1. What is a chemical property? Give examples based on the images provided.
Background
Topic: Chemical Properties of Matter
This question is testing your understanding of chemical properties, which are characteristics of a substance that become evident during or after a chemical reaction. Chemical properties describe how a substance interacts with other substances and the types of chemical changes it can undergo.
Key Terms and Concepts:
Chemical Property: A characteristic that can only be observed or measured when a substance undergoes a chemical change (reaction).
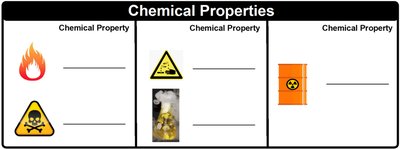
Examples: Flammability, toxicity, reactivity with acids, and radioactivity.
Step-by-Step Guidance
Review the definition of a chemical property and how it differs from a physical property. Chemical properties involve a change in the substance's chemical identity.
Look at the images provided: the flame (flammability), the skull and crossbones (toxicity), the beaker with fumes (reactivity), and the radioactive symbol (radioactivity). Think about what each symbol represents in terms of chemical behavior.
For each image, identify the chemical property being illustrated. For example, flammability is the ability of a substance to burn in the presence of oxygen, while toxicity refers to the harmful effects a substance can have on living organisms.
Write a brief description of each chemical property shown, making sure to connect it to the concept of chemical change (i.e., the substance must undergo a chemical reaction for the property to be observed).
Try solving on your own before revealing the answer!
Final Answer:
Examples of chemical properties include:
Flammability (ability to burn)
Toxicity (harmful effects on organisms)
Reactivity (how a substance reacts with others, such as acids or bases)
Radioactivity (emission of radiation as a result of nuclear decay)
These properties can only be observed when the substance undergoes a chemical change.
