 Back
BackFundamental Concepts in General Chemistry: Atoms, Elements, Bonding, Water, and Chemical Equations
Study Guide - Smart Notes

Atoms and Atomic Structure
Definition and Components of the Atom
An atom is the smallest unit of an element that retains the properties of that element. Atoms are composed of subatomic particles arranged in a specific structure.
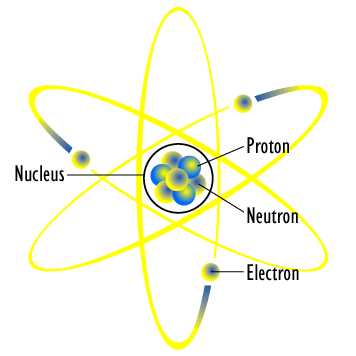
Nucleus: The dense central core of the atom, containing protons and neutrons.
Proton (p+): Positively charged particle found in the nucleus.
Neutron (n0): Neutral particle found in the nucleus.
Electron (e-): Negatively charged particle found in regions (energy levels) surrounding the nucleus.
Energy Levels and Electron Arrangement
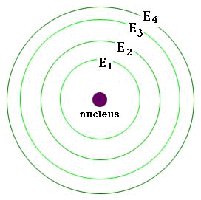
Electrons occupy specific energy levels (also called shells) around the nucleus. Each level can hold a maximum number of electrons:
First energy level: 2 electrons
Second energy level: 8 electrons
Third energy level: 18 electrons (for main group elements, typically 8 for stability)
All occupied energy levels must be filled for an atom to be stable (noble gas configuration).
Electron Configuration
Electron configuration describes the arrangement of electrons in an atom's energy levels.
Example: For an atom with 10 electrons (Neon), the configuration is 2 in the first level, 8 in the second. This is stable.
For an atom with 16 electrons (Sulfur), the configuration is 2 in the first, 8 in the second, 6 in the third. This is not a full outer shell, so it is not stable.
Elements and the Periodic Table
Definition and Classification
An element is a pure substance that cannot be broken down into simpler substances by chemical means. Each element is defined by its number of protons (atomic number).
There are 90 naturally occurring elements.
All elements are organized in the Periodic Table.
Only 25 elements are essential for life; C, H, N, and O make up 96% of human body mass.
Atomic Number and Atomic Mass
Atomic Number (Z): Number of protons in the nucleus; also equals the number of electrons in a neutral atom.
Atomic Mass (A): Sum of protons and neutrons in the nucleus.
Determining Subatomic Particles
Number of protons = atomic number
Number of electrons = atomic number (for neutral atoms)
Number of neutrons = atomic mass - atomic number
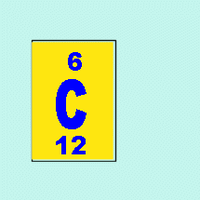
Example: Carbon (atomic number 6, atomic mass 12): 6 protons, 6 electrons, 6 neutrons
Ions and Isotopes
Ions: Atoms with a net charge due to loss or gain of electrons.
Positive ions (cations): Fewer electrons than protons.
Negative ions (anions): More electrons than protons.
Isotopes: Atoms of the same element with different numbers of neutrons (e.g., Carbon-12, Carbon-13, Carbon-14).
Chemical Bonding
How Elements Combine
Compound: Substance formed when two or more different elements bond together (e.g., NaCl, H2O).
Molecule: Group of atoms held together by covalent bonds (e.g., O2).
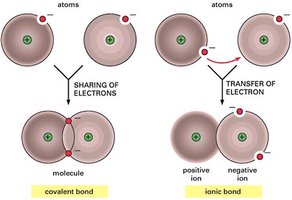
Covalent Bonds
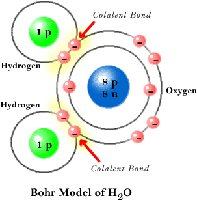
Covalent bonds form when two atoms share electrons. These are common in organic compounds.
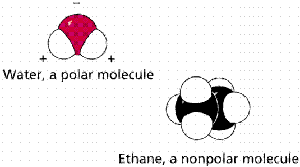
Polar covalent bond: Electrons are shared unequally, resulting in partial charges (e.g., H2O).
Nonpolar covalent bond: Electrons are shared equally (e.g., H2).
Ionic Bonds
Ionic bonds form when one atom transfers electrons to another, resulting in oppositely charged ions that attract each other (e.g., Na+ + Cl- → NaCl).
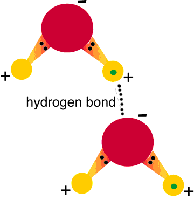
Hydrogen Bonds
Hydrogen bonds are weak attractions between a hydrogen atom in one molecule and an electronegative atom (like oxygen or nitrogen) in another. They are crucial for the structure of water and biological macromolecules.
Water: Structure, Properties, and Importance
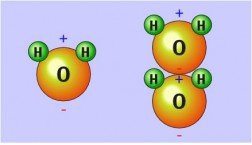
Structure and Polarity of Water
Water (H2O) is a polar molecule due to the unequal sharing of electrons between oxygen and hydrogen. Oxygen is more electronegative, giving it a partial negative charge, while hydrogens are partially positive.
Properties of Water
Cohesion: Attraction between water molecules due to hydrogen bonding (responsible for surface tension).
Adhesion: Attraction between water molecules and other substances (causes meniscus and capillary action).
High Specific Heat Capacity: Water absorbs large amounts of heat before changing temperature, stabilizing environments.
Evaporative Cooling: As water evaporates, it removes heat (e.g., sweating).
Versatile Solvent: Water dissolves many substances due to its polarity.

Solutions and pH
Solutions
A solution consists of a solute (substance dissolved) and a solvent (substance doing the dissolving).
Water is known as the "universal solvent" due to its ability to dissolve many substances.
pH: Acids and Bases
The pH scale measures the concentration of hydrogen ions (H+) in a solution, ranging from 0 (most acidic) to 14 (most basic).
Acid: Substance with more H+ ions (pH < 7); e.g., HCl in water.
Base: Substance with more OH- ions (pH > 7); e.g., NaOH in water.
Neutral: Equal H+ and OH- ions (pH = 7); e.g., pure water.



Chemical Equations and Stoichiometry
Writing and Interpreting Chemical Equations
Chemical equations represent the transformation of reactants into products.
Reactants: Substances present before the reaction.
Products: Substances formed by the reaction.
Chemical equation example:
Coefficients: Numbers before compounds/elements indicating the number of molecules or moles (e.g., 6CO2 means 6 molecules of CO2).
Subscripts: Numbers within formulas indicating the number of atoms in a molecule (e.g., H2O has 2 hydrogens per molecule).
Summary Table: Key Atomic and Chemical Properties
Term | Definition | Example |
|---|---|---|
Atom | Smallest unit of an element | Carbon atom (C) |
Element | Substance with only one type of atom | Oxygen (O2) |
Compound | Substance of two or more elements chemically bonded | Water (H2O) |
Molecule | Group of atoms bonded covalently | Oxygen (O2) |
Covalent Bond | Bond formed by sharing electrons | H2O |
Ionic Bond | Bond formed by transfer of electrons | NaCl |
Hydrogen Bond | Weak attraction between polar molecules | Between H2O molecules |
