 Back
BackFoundations of General Chemistry for Organic Chemistry
Study Guide - Smart Notes

Introduction to Organic Chemistry
Definition and Scope
Organic chemistry is the branch of chemistry that studies the structure, properties, composition, reactions, and synthesis of carbon-containing compounds, which are typically found in biological systems. Organic molecules are essential to life and are found in everyday products such as hairspray, medicines, and nerve agents.
Organic Molecule: Any molecule containing both carbon (C) and hydrogen (H).
Hydrocarbon: An organic molecule composed solely of carbon and hydrogen atoms.
Application: Identifying organic and hydrocarbon molecules in product ingredient lists.

Atoms and Atomic Structure
Basic Atomic Structure
The atom is the fundamental unit of matter, consisting of a nucleus (protons and neutrons) surrounded by electrons.
Atomic Number (Z): Number of protons in the nucleus.
Mass Number (A): Sum of protons and neutrons.
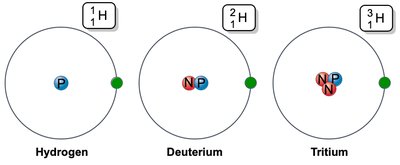
Isotopes: Atoms with the same number of protons but different numbers of neutrons.
Electrons: Negatively charged particles orbiting the nucleus in defined regions called shells and orbitals.
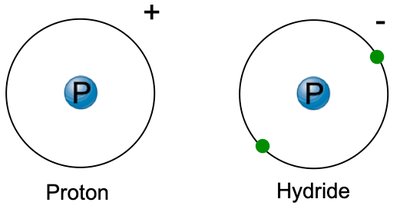
Ions: Atoms with unequal numbers of protons and electrons. Cations are positively charged; anions are negatively charged.
Electron Configuration Principles
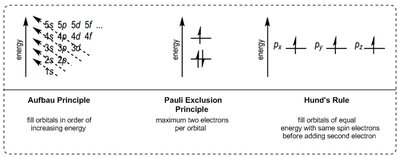
Electron configuration describes the arrangement of electrons in an atom's orbitals, governed by three main principles:
Aufbau Principle: Electrons fill orbitals starting with the lowest energy first.
Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons with opposite spins.
Hund's Rule: Electrons occupy degenerate orbitals singly before pairing up.
Quantum Mechanics and Atomic Orbitals
Wave-Particle Duality and the Heisenberg Uncertainty Principle
Quantum mechanics describes electrons as both particles and waves. The Heisenberg Uncertainty Principle states that it is impossible to know both the position and momentum of an electron simultaneously.
Wave Function (ψ): Mathematical description of the electron's state.
Probability Density: The likelihood of finding an electron in a particular region, derived from the square of the wave function (ψ²).
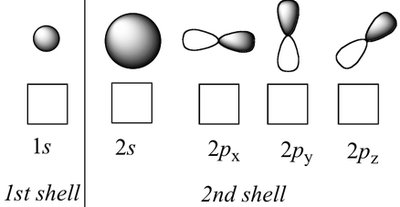
Atomic Orbital: A region in space where there is a high probability of finding an electron.
Electron Interference and Molecular Orbitals
When atomic orbitals overlap, their wave functions can interfere constructively (bonding) or destructively (antibonding), forming molecular orbitals.
Constructive Interference: Leads to bonding molecular orbitals with increased electron density between nuclei.
Destructive Interference: Leads to antibonding molecular orbitals with a node (region of zero electron density).
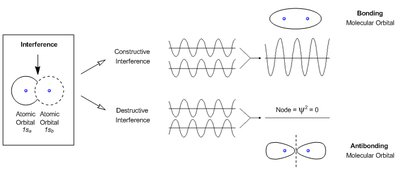
Molecular Orbital Theory
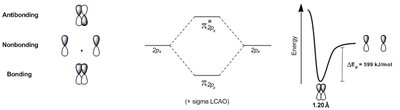
Bonding and Antibonding Orbitals
Molecular Orbital (MO) Theory explains chemical bonding by combining atomic orbitals to form molecular orbitals that extend over the entire molecule.
Bonding Orbital: Lower energy, increased electron density between nuclei.
Antibonding Orbital: Higher energy, node between nuclei.
Linear Combination of Atomic Orbitals (LCAO): Mathematical method to combine atomic orbitals.
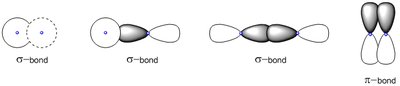
Sigma (σ) and Pi (π) Bonds
Covalent bonds are classified based on the type of orbital overlap:
Sigma (σ) Bond: Formed by head-on overlap of orbitals; allows free rotation.
Pi (π) Bond: Formed by side-to-side overlap of p orbitals; restricts rotation.
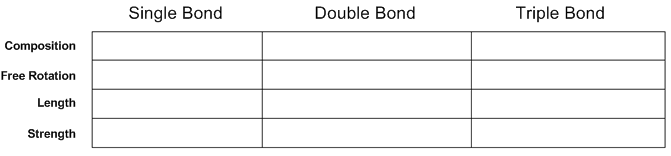
Comparison of Single, Double, and Triple Bonds
The properties of chemical bonds depend on their composition and type:
Single Bond | Double Bond | Triple Bond | |
|---|---|---|---|
Composition | 1 σ | 1 σ + 1 π | 1 σ + 2 π |
Free Rotation | Yes | No | No |
Length | Longest | Intermediate | Shortest |
Strength | Weakest | Intermediate | Strongest |
Octet Rule and Bonding Preferences
Octet Rule
Atoms tend to gain, lose, or share electrons to achieve a noble gas electron configuration, typically eight valence electrons (octet).
First-row elements: H (2 electrons), He (2 electrons), Li (2 electrons).
Second-row elements: C, N, O, F (8 electrons).
Exceptions: Be (4 electrons), B (6 electrons), third-row elements can have expanded octets.
Bonding Preferences by Element
The number of bonds and lone pairs preferred by main group elements is determined by their group number.
Element | Group | Bonding Preference | Bonds | Lone Pairs |
|---|---|---|---|---|
Hydrogen (H) | 1A | 1 bond | 1 | 0 |
Beryllium (Be) | 2A | 2 bonds | 2 | 0 |
Boron (B) | 3A | 3 bonds | 3 | 0 |
Carbon (C) | 4A | 4 bonds | 4 | 0 |
Nitrogen (N) | 5A | 3 bonds | 3 | 1 |
Oxygen (O) | 6A | 2 bonds | 2 | 2 |
Fluorine (F) | 7A | 1 bond | 1 | 3 |
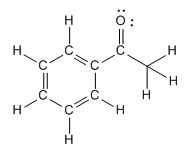
Formal Charges and Stability
Calculating Formal Charge
Formal charge helps determine the most stable Lewis structure for a molecule.
Formula:
Net Charge: The sum of all formal charges in a molecule.
Stability: Structures with formal charges closest to zero are generally more stable.
Bondline (Skeletal) Structures
Drawing and Interpreting Bondline Structures
Bondline (skeletal) structures are simplified representations of organic molecules, where carbon atoms are implied at the ends and intersections of lines, and hydrogens attached to carbons are usually omitted.
Implied Carbons: Each vertex or line end represents a carbon atom.
Implied Hydrogens: Each carbon is assumed to have enough hydrogens to complete its octet.
Heteroatoms: Atoms other than carbon and hydrogen are shown explicitly, with their lone pairs often omitted for simplicity.
Explicit Hydrogens: Hydrogens attached to heteroatoms (e.g., O, N) must be drawn.
Summary Table: Key Concepts
Concept | Definition | Example/Application |
|---|---|---|
Organic Molecule | Contains C and H | Hydrocarbons in fuels |
Atomic Number | Number of protons | Carbon: Z = 6 |
Isotope | Same Z, different neutrons | Hydrogen, Deuterium, Tritium |
Electron Configuration | Arrangement of electrons | 1s2 2s2 2p2 for C |
Octet Rule | 8 valence electrons for stability | Neon, Methane |
Formal Charge | Group # - Valence electrons owned | O in H2O: 6 - (4 + 2) = 0 |
