 Back
BackElements, Compounds, and the Laws of Chemical Combination: Structured Study Notes
Study Guide - Smart Notes

Law of Conservation of Mass
The Law of Conservation of Mass states that in a chemical reaction, matter is neither created nor destroyed, but only changes form. This principle, originated by Antoine Lavoisier, is fundamental to understanding chemical equations and reactions.
Reactants are the substances present before the reaction (left side of the equation).
Products are the substances formed after the reaction (right side of the equation).
The total mass of reactants equals the total mass of products.
Example:
chemical compound always contains the same elements in the same proportion by mass, regardless of the sample size or source.
Mass ratio: Proportion of elements by mass in a compound.
Example: In CO2, the mass ratio of C to O is always constant.
To find unknown mass: Use proportions based on known mass ratios.
Example: If a sample of NaOH contains 10.41 g Na and 0.456 g H, you can calculate the mass of O in another sample using the same ratio.
Atomic Theory
Historical Development and Modern Postulates
The concept of the atom originated with Democritus and was modernized by John Dalton. Dalton's Atomic Theory laid the foundation for understanding matter at the atomic level.
Atoms are indivisible particles (originally stated).
All matter is made of atoms.
Atoms of a given element are identical in mass and properties (modern theory recognizes isotopes).
Atoms combine in simple, whole-number ratios to form compounds.
Chemical reactions rearrange atoms.
Modern updates: Atoms can have different masses (isotopes), and not all combine in simple ratios.
s s | |
|---|---|
Law of Multiple Proportions
Concept and Example
The Law of Multiple Proportions states that when two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
Example: CO and CO2 have mass ratios of O to C that are simple multiples.
Steps: Find atomic masses, calculate mass ratios, compare ratios for whole numbers.
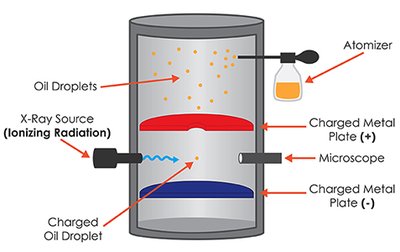
Millikan Oil Drop Experiment
Discovery of Electron Charge
The Millikan Oil Drop Experiment determined the fundamental charge of the electron. Oil droplets were suspended between charged plates, and their behavior under electric and gravitational forces was observed.
Atomizer disperses oil droplets.
X-ray source ionizes droplets.
Charged plates create an electric field.
Balance of electric and gravitational forces allows calculation of electron charge.
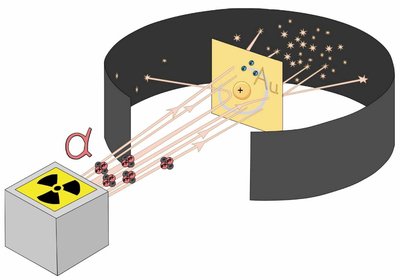
Rutherford Gold Foil Experiment
Discovery of the Atomic Nucleus
Ernest Rutherford's Gold Foil Experiment revealed the existence of a dense, positively charged nucleus at the center of the atom. Alpha particles were directed at thin gold foil, and their scattering patterns were observed.
Most alpha particles passed through, but some were deflected, indicating a small, dense nucleus.
Disproved the Plum Pudding Model.
Nucleus contains protons and neutrons; electrons surround the nucleus.
The Atom and Subatomic Particles
Structure and Properties
An atom consists of a nucleus (protons and neutrons) and electrons orbiting around it. Subatomic particles differ in mass and charge.
Protons: Positive charge, found in nucleus.
Neutrons: Neutral charge, found in nucleus.
Electrons: Negative charge, orbit nucleus.
1 amu = kg
Isotopes
Definition and Notation
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Atomic Number (Z): Number of protons.
Mass Number (A): Number of protons + neutrons.
Isotope notation:
For neutral atoms: Number of protons = number of electrons.
Ions
Cation and Anion Formation
Ions are formed when atoms gain or lose electrons.
Cation: Positive ion formed by loss of electrons.
Anion: Negative ion formed by gain of electrons.
Isoelectronic: Species with the same number of electrons.
Atomic Mass
Calculation and Significance
Atomic mass is the weighted average mass of an element's isotopes, measured in amu, Da, or kg.
Atomic mass formula:
Fractional abundance = percent abundance / 100
Periodic Table: Classifications
Metals, Non-metals, and Metalloids
The Periodic Table classifies elements as metals, non-metals, or metalloids based on their physical and chemical properties.
Metals: Largest group, good conductors, malleable, ductile.
Non-metals: Poor conductors, brittle, opposite properties to metals.
Metalloids: Intermediate properties, border between metals and non-metals.

Periodic Table: Group Names and Organization
Periods, Groups, and Representative Elements
Elements are organized into periods (horizontal rows) and groups (vertical columns). Groups may have unique names and similar chemical properties.
Transition metals: Groups 3-12, variable charges.
Representative elements: Groups 1, 2, and 13-18.
Periodic Table: Element Symbols and Forms
Symbols and Natural Forms
Element symbols are abbreviations, often based on English or Latin names. Elements exist as monoatomic, diatomic, or polyatomic forms in nature.
Monoatomic: Stable as single atoms (e.g., noble gases).
Diatomic: Stable as pairs (e.g., H2, O2, N2).
Polyatomic: Stable as groups larger than two (e.g., S8).
Periodic Table: Phases
States of Matter at Room Temperature
Elements can exist as solids, liquids, or gases at standard room temperature (25°C) and pressure (1 atm).
Solids: Fixed shape and volume.
Liquids: Fixed volume, shape of container.
Gases: Shape and volume of container.

Periodic Table: Charges
Ion Formation and Charge Prediction
Elements form ions to achieve stable electron configurations similar to noble gases.
Metals: Lose electrons, form cations.
Non-metals: Gain electrons, form anions.
Type I Metals: Fixed charge.
Type II Metals: Variable charge (transition metals).
Polyatomic Ions
Structure and Naming
Polyatomic ions are groups of atoms with an overall charge, often containing oxygen (oxyanions).
Oxyanions: Negatively charged, names depend on number of oxygens (e.g., nitrate, nitrite).
Halogen oxyanions: Contain halogens, charge usually -1.
Polyatomic cations: NH4+ (ammonium), Hg22+ (mercury(I)).

Naming Ionic Compounds
Rules and Examples
Ionic compounds consist of a cation (metal or polyatomic ion) and an anion (nonmetal or polyatomic ion).
Cation written first, keeps its name.
If metal has variable charge, use Roman numerals.
Anion changes ending to -ide.
Polyatomic ions keep their names.
Writing Ionic Compounds
Formula Construction
To write ionic compound formulas, balance the charges of the ions involved.
If charges are equal, combine elements directly.
If charges differ, use subscripts to balance.
Naming Ionic Hydrates
Rules and Prefixes
Ionic hydrates are compounds linked to water molecules. The number of water molecules is indicated by numerical prefixes (mono-, di-, tri-, etc.).
Naming Acids
Binary Acids and Oxyacids
Acids are covalent compounds beginning with hydrogen.
Binary acids: H+ bonded to a nonmetal anion (not oxygen).
Prefix "hydro-", suffix "-ic acid".
Oxyacids: H+ bonded to a polyatomic ion containing oxygen.
If ion ends in -ate, acid ends in -ic; if -ite, acid ends in -ous.
Naming Molecular Compounds
Rules and Prefixes
Molecular (covalent) compounds contain only nonmetals. Numerical prefixes indicate the number of each element.
First nonmetal named normally, no "mono-" prefix.
Second nonmetal uses prefix and ends in -ide.
If prefix ends in "a" and next element starts with "o", drop the "a" (e.g., tetroxide).
Numerical Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-
