 Back
BackChapter 5: Thermochemistry and Chemical Energy – Study Notes
Study Guide - Smart Notes

Thermochemistry and Chemical Energy
Introduction to Thermochemistry
Thermochemistry is the study of the energy and heat associated with chemical reactions and physical transformations. It is a branch of chemical thermodynamics, focusing on the changes in energy, particularly heat, during chemical processes.
System: The part of the universe being studied (e.g., the contents of a reaction vessel).
Surroundings: Everything outside the system.
State Functions: Properties that depend only on the state of the system, not on how it got there (e.g., internal energy, enthalpy).
Chemical Energy and Electrostatic Potential
Electrostatic Potential Energy
Electrostatic potential energy is the energy resulting from the interaction between charged particles. It is a key concept in understanding the forces that hold atoms and molecules together.
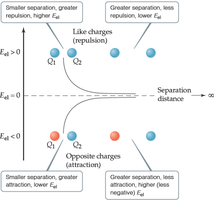
Formula: , where k is a constant, Q1 and Q2 are charges, and d is the distance between them.
Significance: Opposite charges yield negative (attraction), like charges yield positive $E_{el}$ (repulsion).
Example: Two particles of opposite charges always have negative electrostatic potential energy, regardless of distance.

Internal Energy, Heat, and Work
Internal Energy (), Heat (), and Work ()
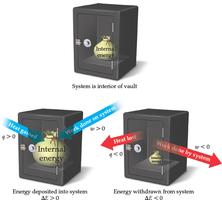
Internal energy is the total energy contained within a system. Changes in internal energy can occur through heat exchange or work done by/on the system.
First Law of Thermodynamics:
Sign Conventions:
: Heat absorbed by the system (endothermic)
: Heat released by the system (exothermic)
: Work done on the system
: Work done by the system
Example: If a system releases 200 kJ of heat and does 35 kJ of work on the surroundings, kJ, kJ, kJ.
Enthalpy and Calorimetry
Enthalpy ()
Enthalpy is a state function that reflects the heat content of a system at constant pressure. It is especially useful for describing heat changes in chemical reactions.
Formula:
Exothermic: (heat released)
Endothermic: (heat absorbed)
Extensive Property: Enthalpy depends on the amount of substance (stoichiometry matters).
Calorimetry
Calorimetry is the measurement of heat flow in a chemical reaction. At constant pressure, the heat measured is equal to the enthalpy change.
Formula:
Heat Capacity (C): The amount of heat required to raise the temperature of an object by 1°C.
Specific Heat (c): The amount of heat required to raise the temperature of 1 gram of a substance by 1°C.
Example: Mixing 50.0 mL of 1.0 M HCl and 50.0 mL of 1.0 M NaOH increases temperature from 21.0°C to 27.5°C. ; .
Hess's Law
Hess's Law of Heat Summation
Hess's Law states that the total enthalpy change for a reaction is the same, no matter how many steps the reaction is carried out in. This allows calculation of enthalpy changes for reactions that are difficult to measure directly.
Formula:
Application: Used to determine enthalpy changes using known enthalpies of formation or reaction steps.
Standard Enthalpy of Formation
Definition and Calculation
The standard enthalpy of formation () is the enthalpy change when one mole of a compound is formed from its elements in their standard states.
Standard State: The most stable physical form of an element or compound at 1 atm and 25°C.
For Elements: for elements in their standard state (e.g., , ).
Example:
Bond Enthalpy and Estimating Reaction Enthalpy
Bond Enthalpy (Bond Energy)
Bond enthalpy is the energy required to break one mole of a particular type of bond in a gaseous molecule. It is used to estimate the enthalpy change of reactions.
Formula:
Example 1: kJ
Example 2: kJ
Key Equations and Constants
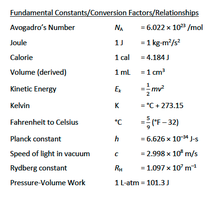
Fundamental Constants and Relationships
Avogadro's Number:
Joule:
Calorie:
Volume:
Kinetic Energy:
Kelvin:
Fahrenheit to Celsius:
Planck constant:
Speed of light:
Rydberg constant:
Pressure-Volume Work:

Common Equations in Thermochemistry
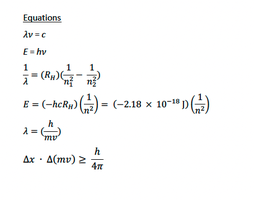
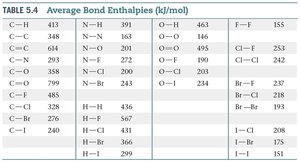
Summary Table: Average Bond Enthalpies (kJ/mol)
The following table provides average bond enthalpies for common bonds, useful for estimating reaction enthalpies:
Bond | Bond Enthalpy (kJ/mol) |
|---|---|
C—H | 413 |
H—H | 436 |
O=O | 495 |
H—Cl | 431 |
F—F | 155 |
Cl—Cl | 242 |
H—F | 567 |
O—H | 463 |
Additional info: | See full table in image_9 for more bond types. |
Practice and Application
Apply sign conventions for and to determine energy changes in systems and surroundings.
Use calorimetry data to calculate enthalpy changes per mole of reactant.
Write standard enthalpy of formation reactions using elements in their standard states.
Estimate reaction enthalpy using bond enthalpy tables and stoichiometric coefficients.
Additional info:
Some equations and constants provided are also relevant for atomic structure and quantum chemistry (see image_3), but are included here for completeness as they may be referenced in thermochemistry problems.
