 Back
BackCarbon and Biological Macromolecules: Structure, Function, and Chemistry
Study Guide - Smart Notes

Carbon: The Foundation of Organic Molecules
Unique Properties of Carbon
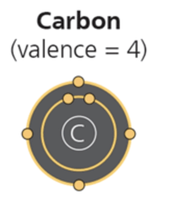
Carbon is the central element in organic chemistry due to its ability to form four covalent bonds, allowing for a diversity of stable and complex molecules essential for life.
Valence Electrons: Carbon has four valence electrons, enabling it to form up to four covalent bonds with other atoms.
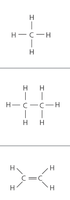
Versatility: Carbon can form single, double, or triple bonds and can bond with many elements, including hydrogen, oxygen, nitrogen, and other carbons.
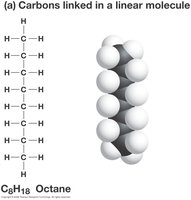
Hydrocarbons: Chains or rings of carbon and hydrogen form the backbone of most organic molecules.
Chemical Groups and Molecular Diversity
The properties of organic molecules are largely determined by the chemical groups attached to their carbon skeletons. These functional groups influence molecular shape, reactivity, and solubility.
Hydroxyl (-OH): Alcohols; increase solubility in water.
Carbonyl (>C=O): Found in aldehydes and ketones; increases reactivity.
Carboxyl (-COOH): Organic acids; can donate protons (H+).
Amino (-NH2): Amines; act as bases.
Sulfhydryl (-SH): Thiols; can form disulfide bonds in proteins.
Phosphate (-PO4): Organic phosphates; involved in energy transfer.

Macromolecules: The Building Blocks of Life
Types and Formation of Macromolecules
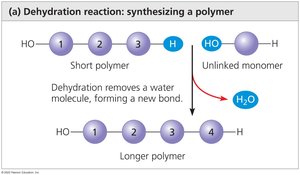
Macromolecules are large, complex molecules essential for life, including carbohydrates, lipids, proteins, and nucleic acids. Most are polymers, made by linking monomers through covalent bonds.
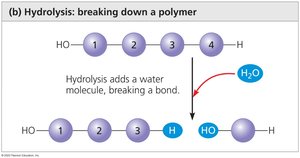
Polymerization (Dehydration Synthesis): Monomers are joined by removing a water molecule, forming a covalent bond.
Hydrolysis: Polymers are broken down into monomers by adding water, breaking covalent bonds.
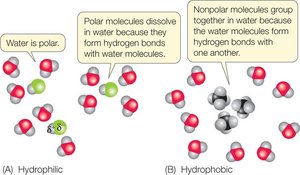
Hydrophobic, Hydrophilic, and Amphipathic Molecules
The solubility of molecules in water depends on their polarity:
Hydrophobic: Non-polar molecules that do not dissolve in water (e.g., many lipids).
Hydrophilic: Polar or charged molecules that dissolve in water (e.g., sugars, salts).
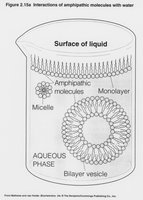
Amphipathic: Molecules with both hydrophobic and hydrophilic regions (e.g., phospholipids).
Carbohydrates: Structure and Function
Classification and Functions
Carbohydrates are essential for energy storage and structural support. They are classified by size:
Monosaccharides: Simple sugars (e.g., glucose, fructose) with the formula .
Disaccharides: Two monosaccharides joined by a glycosidic bond (e.g., sucrose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose).
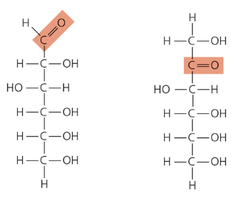
Monosaccharide Structure
Contain a carbonyl group (>C=O) and multiple hydroxyl groups (-OH).
Can exist in linear or ring forms; ring form predominates in aqueous solution.
Classified as aldoses (carbonyl at end) or ketoses (carbonyl in middle).

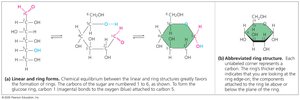
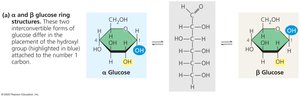
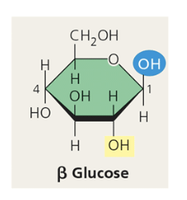
Polysaccharides
Energy Storage: Starch (plants) and glycogen (animals) are storage polysaccharides.
Structural: Cellulose (plants) provides structural support; its parallel chains are held together by hydrogen bonds.
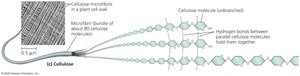
Proteins: Structure and Function
Amino Acids and Peptide Bonds
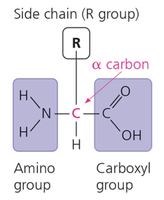
Proteins are polymers of amino acids, joined by peptide bonds. Each amino acid has a central (alpha) carbon, an amino group, a carboxyl group, a hydrogen atom, and a variable side chain (R group).
Peptide Bond Formation: Dehydration synthesis links amino acids; hydrolysis breaks them apart.
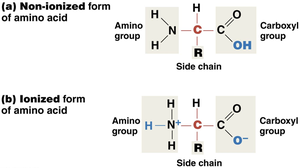
Ionization: In water, amino acids often exist in ionized form.
Classification of Amino Acids
Non-polar (hydrophobic): Side chains mostly C and H.
Polar (hydrophilic): Side chains contain O or N, not charged.
Electrically Charged (hydrophilic): Side chains are acidic (negative) or basic (positive).

Levels of Protein Structure
Primary: Sequence of amino acids, held by peptide bonds.
Secondary: Local folding (alpha helix, beta sheet) via hydrogen bonds in the backbone.
Tertiary: 3D shape from side chain interactions (hydrogen, ionic, disulfide bonds).
Quaternary: Association of multiple polypeptide chains.
Lipids: Structure and Types
Major Classes of Lipids
Triacylglycerols (Triglycerides): Glycerol + 3 fatty acids; energy storage.
Steroids: Four fused hydrocarbon rings; hormones and membrane components.
Phospholipids: Glycerol + 2 fatty acids + phosphate group; amphipathic, major component of cell membranes.
Nucleic Acids: DNA and RNA
Structure and Function
Monomers: Nucleotides (phosphate group, five-carbon sugar, nitrogenous base).
DNA: Double helix, deoxyribose sugar, bases A, T, C, G.
RNA: Single strand, ribose sugar, bases A, U, C, G.
Base Pairing: DNA: A-T, C-G; RNA: A-U, C-G.
Polymerization: Nucleotides linked by phosphodiester bonds (5' to 3' direction).
Summary Table: Macromolecule Types and Functions
Macromolecule | Monomer | Main Functions |
|---|---|---|
Carbohydrates | Monosaccharides | Energy storage, structure |
Proteins | Amino acids | Structure, catalysis, transport, signaling |
Lipids | Fatty acids, glycerol (not true polymers) | Energy storage, membranes, signaling |
Nucleic Acids | Nucleotides | Genetic information storage and transfer |
