 Back
BackAtoms, Elements, and Chemical Bonding: Foundations of General Chemistry
Study Guide - Smart Notes

Atoms and Matter
Definition and Structure of Matter
Matter is anything that takes up space and has mass, including all living and nonliving things. All matter is composed of chemical elements, which are pure substances made of only one type of atom. The atom is the smallest unit of an element and, therefore, the smallest unit of matter that retains the properties of that element.
Matter: Anything with mass and volume (e.g., organisms, rocks, water).
Chemical Element: A pure substance consisting of only one kind of atom.
Atom: The smallest unit of an element, composed of subatomic particles.
Subatomic Particles
Atoms are made up of three main subatomic particles: protons, neutrons, and electrons. Each has a characteristic charge, mass, and location within the atom.
Proton: Positively charged, mass of 1 atomic mass unit (AMU), located in the nucleus.
Neutron: No charge (neutral), mass of 1 AMU, located in the nucleus.
Electron: Negatively charged, negligible mass, orbits the nucleus in electron shells.
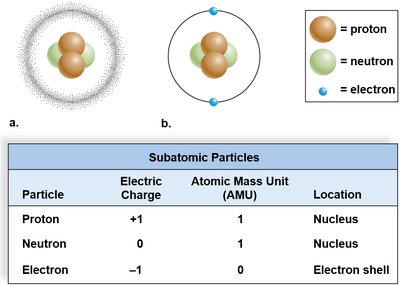
Subatomic Particle | Electric Charge | Atomic Mass Unit (AMU) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | 0 | Electron shell |
Examples of Atoms in Nature
Atoms make up both living and nonliving matter. For example, a diamond is composed entirely of carbon atoms, while glucose (a sugar found in honey) contains carbon, hydrogen, and oxygen atoms.
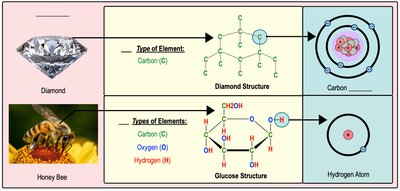
Atomic Structure and Properties
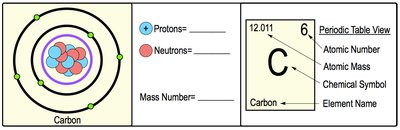
Atomic Number, Mass Number, and Atomic Mass
Each atom of an element has unique properties:
Atomic Number (Z): The number of protons in the nucleus; defines the element.
Mass Number (A): The total number of protons and neutrons in the nucleus.
Atomic Mass: The weighted average mass of all isotopes of an element.
For example, carbon has an atomic number of 6 (6 protons) and a typical mass number of 12 (6 protons + 6 neutrons).
Periodic Table and Elements of Life
The periodic table arranges all known elements based on their chemical properties. Only a small subset of elements is found in living organisms, with about 97% of the mass of most life forms composed of carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur (CHNOPS).
Electron Configuration and Energy Shells
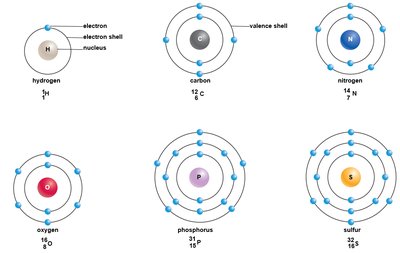
Electron Orbitals and Shells
Electrons occupy three-dimensional regions called orbitals, which are grouped into energy shells around the nucleus. Shells closer to the nucleus are lower in energy, while those farther away are higher in energy. The outermost shell is called the valence shell, and its electrons are called valence electrons.
1st shell: Holds up to 2 electrons
2nd shell: Holds up to 8 electrons
3rd shell: Holds up to 18 electrons (not shown in basic diagrams)
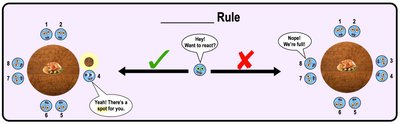
Octet Rule
The octet rule states that atoms are more stable (less reactive) when their valence shells are fully occupied, typically with 8 electrons (except for the first shell, which is full with 2 electrons). Atoms will gain, lose, or share electrons to achieve a full valence shell.

Isotopes and Atomic Mass
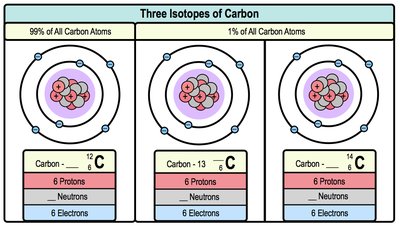
Isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. This results in different mass numbers but the same atomic number. The atomic mass of an element is the weighted average of all its isotopes.
Isotope | Protons | Neutrons | Electrons |
|---|---|---|---|
Carbon-12 | 6 | 6 | 6 |
Carbon-13 | 6 | 7 | 6 |
Carbon-14 | 6 | 8 | 6 |
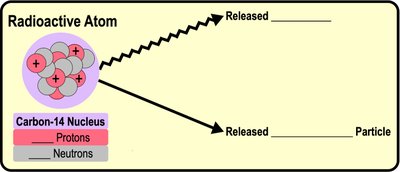
Radioactive Isotopes and Half-Life
Radioactive isotopes are unstable and break down, emitting energy in the form of radiation. The half-life is the time it takes for half of the radioactive atoms in a sample to decay. Radioactive isotopes are used in medicine and for dating fossils.
Chemical Bonding
Types of Chemical Bonds
Chemical bonds are attractive forces that hold atoms together in molecules and compounds. There are two main categories:
Intramolecular Bonds: Bonds within a molecule (e.g., covalent, ionic bonds).
Intermolecular Bonds: Bonds between molecules (e.g., hydrogen bonds, van der Waals forces).
Covalent Bonds
Covalent bonds involve the sharing of electron pairs between atoms. There are two types:
Nonpolar Covalent Bonds: Equal sharing of electrons (atoms have similar electronegativities).
Polar Covalent Bonds: Unequal sharing of electrons (atoms have different electronegativities), resulting in partial charges (δ+ and δ−).
Electronegativity is a measure of an atom's ability to attract shared electrons in a bond (scale: 0–4).
Noncovalent Bonds
Noncovalent bonds are interactions that do not involve sharing of electrons. Types include:
Ionic Bonds: Attraction between oppositely charged ions (cations and anions).
Hydrogen Bonds: Weak attraction between a hydrogen atom covalently bonded to a highly electronegative atom (F, O, or N) and another electronegative atom.
Van der Waals Forces: Weak attractions due to temporary dipoles in molecules.
Ions: Anions and Cations
An ion is an atom or molecule with a net electrical charge due to the loss or gain of electrons.
Anion: Negatively charged ion (gains electrons).
Cation: Positively charged ion (loses electrons).
Ionic Bonding
Ionic bonds are formed by the transfer of electrons from one atom to another, resulting in the formation of oppositely charged ions that attract each other. This transfer allows both atoms to achieve full valence shells.
Hydrogen Bonding
Hydrogen bonds are weak individually but can be strong collectively. They are crucial in determining the properties of water and the structure of biological macromolecules (e.g., DNA, proteins).
Summary Table: Types of Chemical Bonds
Bond Type | Mechanism | Strength | Example |
|---|---|---|---|
Covalent (Nonpolar) | Equal sharing of electrons | Strong | O2, N2 |
Covalent (Polar) | Unequal sharing of electrons | Strong | H2O |
Ionic | Transfer of electrons | Strong (in solid state) | NaCl |
Hydrogen | Attraction between H and electronegative atom | Weak (individually) | Between water molecules |
Van der Waals | Temporary dipole interactions | Very weak | Between nonpolar molecules |
Additional info: This guide covers foundational concepts from Focus 0 (Matter, Measurements, Reactions, and Stoichiometry), Focus 1 (Atomic Structure, Electron Configuration, and Periodic Trends), and Focus 2 (Chemical Bonding and Molecular Shapes) of a general chemistry curriculum.
