 Back
BackAtomic Structure, Bonding, and Chemical Properties: Study Guide
Study Guide - Smart Notes

Bonding and Atomic Structure
Ionic vs. Covalent Bonding
Chemical bonds are the forces that hold atoms together in compounds. The two primary types of chemical bonds are ionic bonds and covalent bonds, which differ in how electrons are distributed between atoms.
Ionic Bonds: Formed when one atom transfers electrons to another, resulting in the formation of oppositely charged ions. Typically occurs between metals and non-metals (e.g., sodium chloride, NaCl).
Covalent Bonds: Formed when two atoms share one or more pairs of electrons. Usually occurs between non-metal atoms (e.g., water, H2O).
Stability: Both types of bonding allow atoms to achieve a stable electron configuration, often resembling that of noble gases.
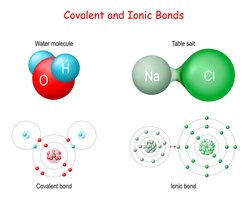
Example: In NaCl, sodium donates an electron to chlorine (ionic bond). In H2O, oxygen shares electrons with hydrogen atoms (covalent bond).
Atomic Properties of Sodium
Understanding atomic structure is fundamental to chemistry. The properties of an element are determined by the number of protons, neutrons, and electrons in its atoms.
Atomic Number: The number of protons in the nucleus. For sodium, the atomic number is 11.
Mass Number: The sum of protons and neutrons. For sodium with 11 protons and 12 neutrons: .
Element Identification: The atomic number uniquely identifies an element. Sodium (Na) always has 11 protons.
Example: An atom with 11 protons and 12 neutrons is sodium-23.
The Stability of Noble Gases
Noble gases (Group 18 elements) are characterized by their lack of chemical reactivity. This is due to their full valence electron shells, which make them energetically stable and unlikely to form bonds.
Valence Shell: The outermost electron shell. Noble gases have 8 electrons in their valence shell (except helium, which has 2).
Chemical Inertness: Their stable configuration means they rarely participate in chemical reactions.
Example: Neon (Ne) and Argon (Ar) are noble gases that do not readily form compounds.
Chemical Reactions and Properties
Balancing Chemical Equations
Balancing chemical equations ensures that the Law of Conservation of Mass is obeyed: atoms are neither created nor destroyed in a chemical reaction.
Unbalanced Equation:
Balanced Equation:
Explanation: Coefficients are used to ensure the same number of each type of atom on both sides of the equation.
Example: In the balanced equation above, there are 4 hydrogen atoms and 2 oxygen atoms on both sides.
Identifying Acids and Bases
The pH scale is used to determine the acidity or basicity of a solution, based on the concentration of hydrogen ions (H+).
Acids: pH less than 7 (0–6), high concentration of H+ ions. Strong acids have pH values close to 0–2.
Bases (Alkaline): pH greater than 7 (8–14), low concentration of H+ ions, high concentration of OH− ions.
Neutral: pH of 7 (pure water).
Indicators: pH indicators change color depending on the acidity or basicity of a solution.
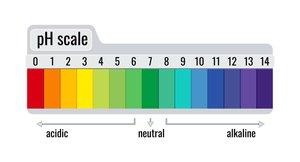
Example: A solution with pH = 2 is a strong acid; a solution with pH = 13 is a strong base.
