 Back
BackAtomic Structure and Nuclear Chemistry: Foundations and Applications
Study Guide - Smart Notes

Atomic Structure and Nuclear Chemistry
Introduction
This section introduces the historical development of atomic theory, the structure of the atom, the concept of isotopes, and the measurement and application of isotopic abundances. These foundational topics are essential for understanding the nature of matter and the principles of nuclear chemistry.
Historical Development of Atomic Theory
Early Ideas about Matter
Ancient Greek Philosophers: Proposed that all matter was composed of four elements: air, earth, fire, and water.
Democritus (~460–370 B.C.): Suggested that matter could be divided until reaching an indivisible particle, called the atom (from Greek: a = not; tomos = cut).
Foundational Laws of Chemistry
Law of Conservation of Mass (Lavoisier, 1785): Mass is neither created nor destroyed in chemical reactions.
Law of Constant Composition (Proust, 1794): A chemical compound always contains the same proportion of elements by mass.
Dalton’s Atomic Theory (1808)
All matter consists of solid, indivisible atoms.
Atoms are indestructible and retain their identity in chemical reactions.
All atoms of a given element are identical in mass and properties.
Atoms of different elements differ in mass and properties.
Compounds are formed by the combination of atoms in small whole-number ratios.
Note: Some postulates have been modified with modern discoveries (e.g., atoms are divisible into subatomic particles).
Structure of the Atom
Subatomic Particles
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles found in the nucleus.
Electrons: Negatively charged particles found in the outer regions of the atom.
Most of the atom's mass is concentrated in the nucleus, which occupies a tiny fraction of the atom's volume.
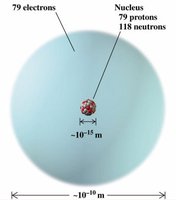
Particle | Mass (kg) | Mass (u) | Charge (C) | Charge (relative) | Location |
|---|---|---|---|---|---|
Electron | 9.109382 × 10-31 | 0.00054858 | -1.602176 × 10-19 | -1 | Outer region |
Proton | 1.672622 × 10-27 | 1.007276 | +1.602176 × 10-19 | +1 | Nucleus |
Neutron | 1.674927 × 10-27 | 1.008665 | 0 | 0 | Nucleus |
Charge of an atom:


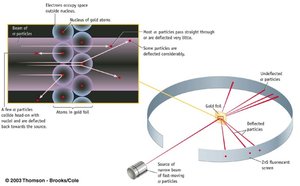
Rutherford’s Gold Foil Experiment
This experiment demonstrated that atoms have a small, dense, positively charged nucleus.
Defining Elements and Isotopes
Atomic Number and Mass Number
Atomic Number (Z): Number of protons in the nucleus; defines the element.
Mass Number (A): Total number of protons and neutrons (nucleons) in the nucleus.
Changing the number of protons changes the element. Atoms of the same element with different numbers of neutrons are called isotopes.
Examples of Isotopes:
, , (Carbon isotopes)
, , (Hydrogen isotopes)
Applications of Isotopes
Dating and Tracing: Isotope ratios are used in biology, geology, paleontology, and archaeology for dating samples (e.g., radiocarbon dating).
Forensic Science: The amount of in tooth enamel can determine the year of birth.
Measuring Isotopes and Atomic Mass
Mass Spectrometry
Mass spectrometry is used to determine the isotopic composition of elements by separating isotopes based on their mass-to-charge ratio.


Weighted Average Atomic Mass
The atomic mass listed on the periodic table is the weighted average of all naturally occurring isotopes of an element.
Example: Silicon
92.23% (27.9769 u)
4.67% (28.9765 u)
3.10% (29.9738 u)
Calculate the average atomic mass using the formula above.
Example: Gallium
Average atomic mass: 69.723 u
: 68.926 u
: 70.925 u
Predict which isotope is more abundant and calculate the natural abundance of each isotope.
Nuclear Chemistry Applications
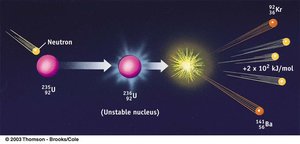
Nuclear Reactions and Energy
Nuclear reactions involve changes in the nucleus and can release large amounts of energy, as seen in fission and fusion processes.
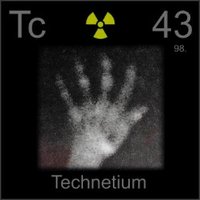
Radioactive Isotopes
Technetium (Tc): An example of a radioactive element used in medical imaging and research.

Applications in Modern Technology
Solar Panels: Use of elements like silicon in photovoltaic cells for energy conversion.

Additional info: The images included illustrate atomic structure, isotopic samples, flame tests for element identification, nuclear reactions, and applications of isotopes in technology and medicine. The table summarizes the properties of subatomic particles. The notes cover the foundational aspects of atomic structure and nuclear chemistry, suitable for introductory college-level general chemistry.
