 Back
BackAtomic Structure and Nuclear Chemistry: Foundations and Applications
Study Guide - Smart Notes

Atomic Structure and Nuclear Chemistry
Historical Development of Atomic Theory
The concept of the atom has evolved over centuries, beginning with philosophical ideas and culminating in scientific theories based on experimental evidence.
Ancient Greek Philosophers: Proposed that all matter was composed of four elements: air, earth, fire, and water. Democritus introduced the idea of the atom as an indivisible particle.
Law of Conservation of Mass (Lavoisier, 1785): Mass is neither created nor destroyed in chemical reactions.
Law of Constant Composition (Proust, 1794): A given compound always contains the same proportion of elements by mass.
Dalton's Atomic Theory (1808):
All matter consists of solid, indivisible atoms.
Atoms are indestructible and retain their identity in chemical reactions.
Atoms of a given element are identical in mass and properties.
Atoms of different elements differ in mass and properties.
Compounds are formed by combinations of atoms in small whole-number ratios.
Note: Modern science has modified some of these postulates, recognizing the existence of subatomic particles and isotopes.
Structure of the Atom
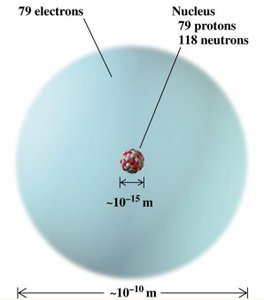
Atoms are composed of three fundamental subatomic particles: protons, neutrons, and electrons. Most of the atom's mass is concentrated in a tiny, dense nucleus.
Protons: Positively charged particles located in the nucleus.
Neutrons: Neutral particles also found in the nucleus.
Electrons: Negatively charged particles that occupy the space around the nucleus.
Most of an atom is empty space. The nucleus is extremely small compared to the overall size of the atom.
Charge of an atom: $\text{Charge} = \text{Number of protons} - \text{Number of electrons}$
Defining an Element and Isotopes
Each element is defined by its atomic number (number of protons). Atoms of the same element can have different numbers of neutrons, resulting in isotopes.
Atomic Number (Z): Number of protons in the nucleus; defines the element.
Mass Number (A): Total number of protons and neutrons (nucleons) in the nucleus.
Isotopes: Atoms of the same element with different numbers of neutrons (and thus different mass numbers).
Changing the number of protons changes the element itself (nuclear reaction).
Example: Carbon has three naturally occurring isotopes: $^{12}\text{C}$, $^{13}\text{C}$, and $^{14}\text{C}$.
Applications of Isotopes
Isotopes have important applications in science and technology, including dating, tracing, and forensic analysis.
Radiocarbon Dating: The ratio of $^{14}\text{C}$ to $^{12}\text{C}$ in biological samples can be used to determine the age of archaeological and geological specimens.
Forensic Science: The amount of $^{14}\text{C}$ in tooth enamel can reveal the year of birth, due to changes in atmospheric $^{14}\text{C}$ from nuclear testing.
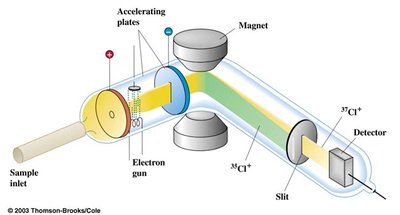
Measuring Isotopes: Mass Spectrometry
Mass spectrometry is a powerful technique used to determine the isotopic composition of elements in a sample.
Principle: Ions of different masses are separated in a magnetic field and detected, producing a spectrum that shows the relative abundance of each isotope.
Main difference between isotopes: Their mass, which allows for separation and identification.
Average Atomic Mass and Isotopic Abundance
Most elements exist as mixtures of isotopes. The atomic mass listed on the periodic table is a weighted average of the masses of all naturally occurring isotopes, based on their relative abundances.
Weighted Average Formula:
$\text{Average atomic mass} = \sum \left( \frac{\text{abundance of isotope}}{100} \times \text{mass of isotope} \right)$
Example (Silicon): Silicon has three naturally occurring isotopes: 92.23% $^{28}\text{Si}$ (27.9769 u), 4.67% $^{29}\text{Si}$ (28.9765 u), and 3.10% $^{30}\text{Si}$ (29.9738 u). The average atomic mass is calculated using the formula above.
Example (Gallium): Gallium has two isotopes, $^{69}\text{Ga}$ and $^{71}\text{Ga}$, with known atomic masses and average atomic mass. The relative abundance of each isotope can be determined algebraically.
Summary Table: Subatomic Particles
Particle | Mass (kg) | Mass (u) | Charge (C) | Charge (relative) | Location |
|---|---|---|---|---|---|
Electron | 9.109382 × 10-31 | 0.00054858 | -1.602176 × 10-19 | -1 | Outer region |
Proton | 1.672622 × 10-27 | 1.007276 | +1.602176 × 10-19 | +1 | Nucleus |
Neutron | 1.674927 × 10-27 | 1.008665 | 0 | 0 | Nucleus |
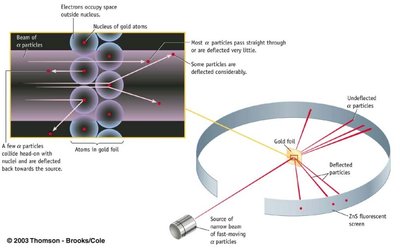
Additional info: The gold foil experiment (Rutherford) provided key evidence for the nuclear model of the atom, showing that most of the atom is empty space and that the nucleus is small and dense.
