 Back
BackOrganic Chemistry Thermodynamics, Acids, Bases, and Reaction Mechanisms – Study Guidance
Study Guide - Smart Notes

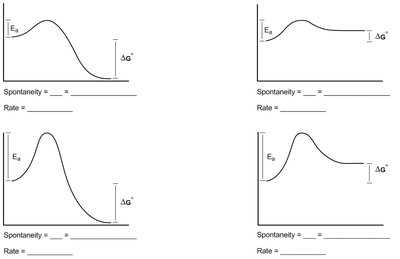
Q1. Describe the favorability and rate of the following free energy diagrams.
Background
Topic: Thermodynamics and Kinetics in Organic Chemistry
This question tests your understanding of how free energy diagrams illustrate the spontaneity (thermodynamic favorability) and rate (kinetic accessibility) of chemical reactions.
Key Terms and Formulas
Gibbs Free Energy (): Determines spontaneity. If , the reaction is spontaneous.
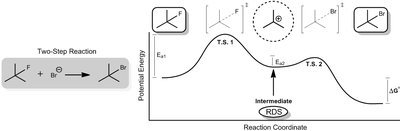
Activation Energy (): Determines the rate. Lower means a faster reaction.
Step-by-Step Guidance
Examine each diagram and identify the relative heights of the reactants and products. The difference in their energy levels represents .
Determine if is negative (products lower than reactants) or positive (products higher than reactants) to assess spontaneity.
Look at the height of the activation energy barrier () for each diagram. A lower barrier means a faster reaction rate.
Compare the diagrams to see which reactions are both thermodynamically favorable and kinetically accessible.
Try solving on your own before revealing the answer!
Final Answer:
For each diagram, spontaneity is determined by the sign of and rate by the magnitude of . Diagrams with are spontaneous, and those with lower proceed faster.
Q2. Explain the three terms that compose Gibbs Free Energy and how each affects reaction favorability.
Background
Topic: Gibbs Free Energy Components
This question focuses on the thermodynamic factors that determine whether a reaction is favorable: enthalpy, entropy, and temperature.
Key Terms and Formulas
Gibbs Free Energy Equation:
= Enthalpy (heat content)
= Entropy (disorder)
= Temperature (in Kelvin)
Step-by-Step Guidance
Recall that represents the sum of bond energies; negative values indicate exothermic reactions (energy released).
measures the change in disorder; positive values mean increased disorder, which favors spontaneity.
Temperature () amplifies the effect of entropy; at higher temperatures, the entropy term becomes more significant in determining favorability.
Combine these terms in the Gibbs equation to predict whether will be negative (favorable) or positive (unfavorable).
Try solving on your own before revealing the answer!
Final Answer:
Gibbs Free Energy is determined by enthalpy, entropy, and temperature. Favorability increases with negative , positive , and higher amplifying entropy's effect.
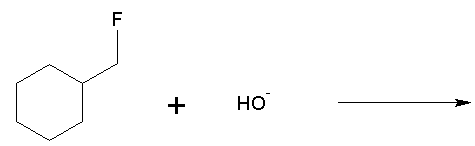
Q3. Identify the type of chemical reaction: acid-base, substitution, elimination, or addition.
Background
Topic: Types of Organic Reactions
This question tests your ability to classify reactions based on the changes in bonds and atoms/groups exchanged.
Key Terms and Formulas
Acid-Base Reaction: Exchange of a proton (H+).
Substitution Reaction: Exchange of an atom/group other than H+.
Elimination Reaction: Removal of two single bonds to form a double bond.
Addition Reaction: Conversion of a double bond to two single bonds.
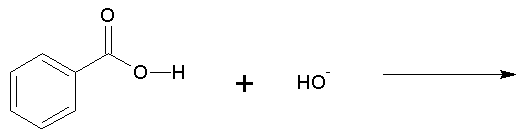

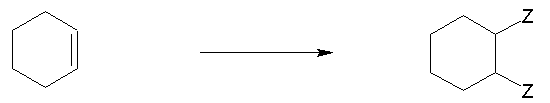
Step-by-Step Guidance
Look at the reactants and products to see if a proton (H+) is exchanged (acid-base).
If an atom/group other than H+ is exchanged, it's a substitution reaction.
If two single bonds are removed and a double bond is formed, it's an elimination reaction.
If a double bond is converted to two single bonds, it's an addition reaction.
Try solving on your own before revealing the answer!
Final Answer:
Each reaction type is identified by the specific bond changes: acid-base (proton exchange), substitution (atom/group exchange), elimination (double bond formation), and addition (double bond consumption).
