 Back
BackWater and Life: Properties, Functions, and Biological Importance
Study Guide - Smart Notes

Ch. 3 - Water & Life
Properties of Water
Water is a unique molecule essential for life, exhibiting several properties that make it vital for biological systems. Its molecular structure and ability to form hydrogen bonds underlie these properties.
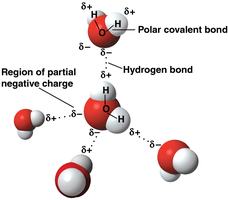
Polarity: Water molecules are polar, with a partial negative charge near the oxygen atom and partial positive charges near the hydrogen atoms.
Hydrogen Bonding: The polarity allows water molecules to form hydrogen bonds with each other and with other polar molecules, contributing to water's cohesion, adhesion, and high surface tension.
Temperature Stabilization: Water can absorb or release large amounts of heat with only slight changes in its own temperature, helping to moderate environmental and organismal temperature.
Temperature and Heat
Water's thermal properties are crucial for maintaining stable conditions in living organisms and environments.
Calorie: A calorie (cal) is the amount of heat required to raise the temperature of 1 g of water by 1ºC.
Specific Heat: Water has a high specific heat (1 cal/g·ºC), meaning it resists temperature changes. This is due to hydrogen bonding, which must be disrupted for temperature to rise.
Biological Impact: Large bodies of water moderate climate by absorbing heat during the day and releasing it at night, stabilizing temperatures for coastal regions and aquatic life.
Evaporative Cooling
Evaporation is a key process by which organisms and environments regulate temperature.
Heat of Vaporization: The heat a liquid must absorb for 1 g to be converted to gas. Water's high heat of vaporization allows for effective cooling.
Evaporative Cooling: As water evaporates, the surface cools because the molecules with the highest kinetic energy leave first. This process helps organisms (e.g., sweating in mammals) and bodies of water maintain stable temperatures.

Floating of Ice on Liquid Water
Water is less dense as a solid than as a liquid, a property with significant ecological consequences.
Crystalline Lattice: At 0ºC, water molecules form a crystalline lattice held together by hydrogen bonds, making ice about 10% less dense than liquid water.
Ecological Impact: Ice floats, insulating the water below and providing a habitat for certain organisms. Water reaches its greatest density at 4ºC.

Water: The Solvent of Life
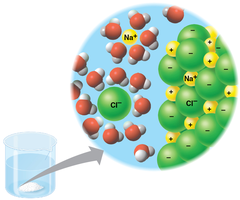
Water's polarity makes it an excellent solvent, capable of dissolving a wide range of substances necessary for life.
Solution: A homogeneous mixture of two or more substances. The solvent is the dissolving agent, and the solute is the substance dissolved.
Aqueous Solution: Water is the solvent.
Hydration Shell: When ionic compounds dissolve, water molecules surround each ion, forming a hydration shell.
Versatility: Water dissolves ionic and many polar molecules, including large molecules like proteins if they have ionic or polar regions.
Hydrophilic vs. Hydrophobic: Hydrophilic substances have an affinity for water; hydrophobic substances do not.
Buffers, pH, Acids, and Bases
The pH of a solution reflects its hydrogen ion concentration and is critical for biological processes. Buffers help maintain pH stability in organisms.
pH Scale: Measures acidity or alkalinity; defined as .
Acids: Donate hydrogen ions (H⁺), lowering pH. Strong acids dissociate completely in water.
Bases: Donate hydroxide ions (OH⁻), raising pH. Strong bases dissociate completely in water.
Neutral Solution: pH = 7; [H⁺] = M at 25ºC.
Buffer Systems: Buffers absorb excess H⁺ or OH⁻ to maintain stable pH. The carbonic acid (H₂CO₃) and bicarbonate (HCO₃⁻) system is crucial in blood and cellular fluids.




Table: Common Substances and Their pH
Substance | pH |
|---|---|
Gastric acid | 1 |
Lemon juice | 2 |
Orange juice | 3 |
Tomato juice | 4 |
Black coffee | 5 |
Urine | 6 |
Distilled water | 7 |
Seawater | 8 |
Baking soda | 9 |
Milk of magnesia | 10 |
Ammonia solution | 11 |
Soapy water | 12 |
Bleach | 13 |
Acidification: A Threat to Our Oceans
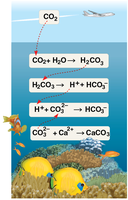
Human activities, especially the burning of fossil fuels, are increasing atmospheric CO₂, a significant portion of which is absorbed by oceans, leading to ocean acidification.
Ocean Acidification: CO₂ dissolves in seawater, forming carbonic acid (H₂CO₃), which dissociates to release H⁺ ions, lowering pH.
Impact on Marine Life: Increased H⁺ combines with carbonate ions (CO₃²⁻), reducing their availability for calcification, which is essential for organisms like corals and shellfish.
Ecological Consequences: Acidification threatens coral reefs and can cause widespread changes in marine ecosystems.
Key Equations
Product of ion concentrations in water:
pH definition:
Carbonic acid-bicarbonate buffer:
