 Back
BackOrganic Chemistry: Thermodynamics, Acids, Bases, and Reaction Mechanisms Study Guidance
Study Guide - Smart Notes

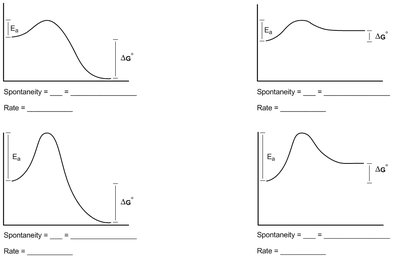
Q1. Describe the favorability and rate of the following free energy diagrams.
Background
Topic: Thermodynamics and Kinetics in Organic Chemistry
This question tests your understanding of how free energy diagrams illustrate the spontaneity (thermodynamic favorability) and rate (kinetic speed) of chemical reactions.
Key Terms and Formulas:
Gibbs Free Energy ($\Delta G^\circ$): Determines spontaneity. If $\Delta G^\circ < 0$, the reaction is spontaneous.
Activation Energy ($E_a$): Determines the rate. Lower $E_a$ means a faster reaction.
Step-by-Step Guidance
Examine each diagram and identify the $\Delta G^\circ$ value. Is it positive or negative? This tells you if the reaction is thermodynamically favorable (spontaneous) or not.
Look at the height of the activation energy barrier ($E_a$) in each diagram. A lower barrier means the reaction will proceed faster; a higher barrier means it will be slower.
Compare the diagrams: Which has the lowest $E_a$? Which has the most negative $\Delta G^\circ$? Use these to rank favorability and rate.
Fill in the blanks for spontaneity and rate based on your analysis of $\Delta G^\circ$ and $E_a$ for each diagram.
Try solving on your own before revealing the answer!
Final Answer:
For each diagram, spontaneity is determined by the sign of $\Delta G^\circ$ (negative = spontaneous), and rate is determined by the size of $E_a$ (smaller = faster). Fill in the blanks accordingly for each diagram.
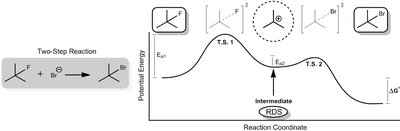
Q2. Explain the significance of transition states and intermediates in multi-step reactions.
Background
Topic: Reaction Mechanisms
This question focuses on the difference between transition states and intermediates in multi-step organic reactions, and how they appear on energy diagrams.
Key Terms and Formulas:
Transition State: A high-energy, unstable configuration where bonds are being broken and formed. Cannot be isolated.
Intermediate: A species formed between steps, more stable than a transition state, and can sometimes be isolated.
Step-by-Step Guidance
Identify the peaks in the energy diagram—these represent transition states (T.S.).
Locate the valleys between peaks—these are intermediates, which are more stable than transition states but less stable than reactants or products.
Understand that the overall $\Delta G^\circ$ for the reaction is the sum of the free energy changes for each step.
Note that the rate-determining step (RDS) is the step with the highest activation energy barrier.
Try solving on your own before revealing the answer!
Final Answer:
Transition states are the highest energy points and cannot be isolated, while intermediates are lower energy and can sometimes be isolated. The RDS is the slowest step, and the overall $\Delta G^\circ$ is the sum of all steps.
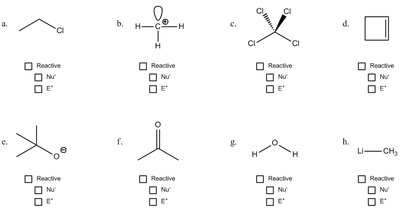
Q3. Identify which of the following molecules would be expected to be reactive, nucleophilic, or electrophilic.
Background
Topic: Reactivity, Nucleophiles, and Electrophiles
This question tests your ability to classify molecules based on their charge and electron density, and predict their behavior in reactions.
Key Terms and Formulas:
Nucleophile: Electron-rich species that donate electrons.
Electrophile: Electron-poor species that accept electrons.
Reactivity Indicators: Charge, lone pairs, bond polarity, and resonance.
Step-by-Step Guidance
Look for negative charges or lone pairs—these often indicate nucleophiles.
Look for positive charges or electron-deficient atoms—these often indicate electrophiles.
Consider resonance and bond polarity to further assess reactivity.
Check each molecule and mark whether it is reactive, nucleophilic, or electrophilic based on these criteria.
Try solving on your own before revealing the answer!
Final Answer:
Negatively charged or electron-rich species are nucleophiles; positively charged or electron-poor species are electrophiles. Mark each molecule accordingly.
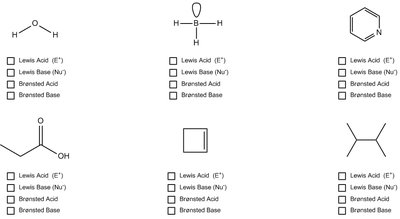
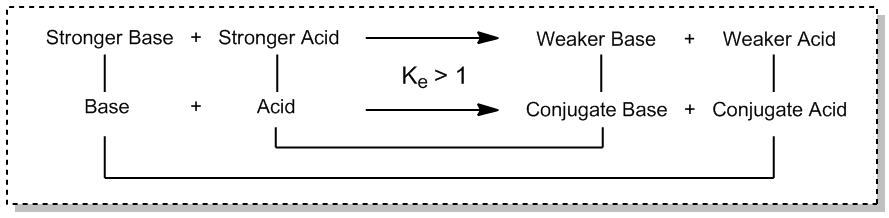
Q4. Properly identify the following molecules as Lewis acid/base or Brønsted acid/base.
Background
Topic: Acid-Base Theory
This question tests your understanding of the Lewis and Brønsted-Lowry definitions of acids and bases, and how to apply them to different molecules.
Key Terms and Formulas:
Lewis Acid: Electron pair acceptor.
Lewis Base: Electron pair donor.
Brønsted Acid: Proton donor.
Brønsted Base: Proton acceptor.
Step-by-Step Guidance
For each molecule, determine if it can donate or accept a proton (Brønsted) or an electron pair (Lewis).
Check for lone pairs (Lewis base) or empty orbitals (Lewis acid).
Assign each molecule to the correct category based on its structure and properties.
Mark the appropriate boxes for each molecule.
Try solving on your own before revealing the answer!
Final Answer:
Classify each molecule as Lewis acid/base or Brønsted acid/base based on its ability to donate/accept protons or electron pairs.
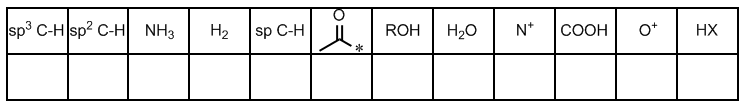
Q5. Rank the following organic compounds in order of increasing acidity and increasing pKa.
Background
Topic: Acid Strength and pKa
This question tests your ability to use pKa values to compare the acidity of different organic compounds.
Key Terms and Formulas:
pKa: A measure of acid strength; lower pKa means stronger acid.
Acidity: The tendency to donate a proton; stronger acids have lower pKa values.
Step-by-Step Guidance
Identify the pKa values for each compound listed.
Arrange the compounds in order of increasing pKa (weakest to strongest acid).
Arrange the compounds in order of increasing acidity (strongest to weakest acid).
Double-check your rankings using the provided pKa table.
Try solving on your own before revealing the answer!
Final Answer:
Rank compounds by pKa (lowest to highest) for increasing acidity, and by acidity (highest to lowest) for increasing pKa.
