Table of contents
- 1. Introduction to Anatomy & Physiology
- What is Anatomy & Physiology?
- Levels of Organization
- Variation in Anatomy & Physiology
- Introduction to Organ Systems
- Homeostasis
- Feedback Loops
- Feedback Loops: Negative Feedback
- Feedback Loops: Positive Feedback
- Anatomical Position
- Introduction to Directional Terms
- Directional Terms: Up and Down
- Directional Terms: Front and Back
- Directional Terms: Body Sides
- Directional Terms: Limbs
- Directional Terms: Depth Within the Body
- Introduction to Anatomical Terms for Body Regions
- Anatomical Terms for the Head and Neck
- Anatomical Terms for the Front of the Trunk
- Anatomical Terms for the Back
- Anatomical Terms for the Arm and Hand
- Anatomical Terms for the Leg and Foot
- Review- Using Anatomical Terms and Directions
- Abdominopelvic Quadrants and Regions
- Anatomical Planes & Sections
- Organization of the Body: Body Cavities
- Organization of the Body: Serous Membranes
- Organization of the Body: Serous Membrane Locations
- Organization of the Body: Thoracic Cavity
- Organization of the Body: Abdominopelvic Cavity
- 2. Cell Chemistry & Cell Components
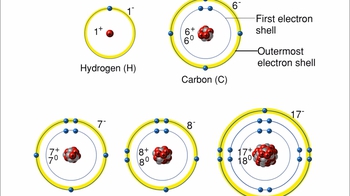
- Atoms- Smallest Unit of Matter
- Isotopes
- Introduction to Chemical Bonding
- Covalent Bonds
- Noncovalent Bonds
- Ionic Bonding
- Hydrogen Bonding
- Introduction to Water
- Properties of Water- Cohesion and Adhesion
- Properties of Water- Density
- Properties of Water- Thermal
- Properties of Water- The Universal Solvent
- Acids and Bases
- pH Scale
- Carbon
- Functional Groups
- Introduction to Biomolecules
- Monomers & Polymers
- Carbohydrates
- Proteins
- Nucleic Acids
- Lipids
- Microscopes
- Prokaryotic & Eukaryotic Cells
- Introduction to Eukaryotic Organelles
- Endomembrane System: Protein Secretion
- Endomembrane System: Digestive Organelles
- Mitochondria & Chloroplasts
- Endosymbiotic Theory
- Introduction to the Cytoskeleton
- Cell Junctions
- Biological Membranes
- Types of Membrane Proteins
- Concentration Gradients and Diffusion
- Introduction to Membrane Transport
- Passive vs. Active Transport
- Osmosis
- Simple and Facilitated Diffusion
- Active Transport
- Endocytosis and Exocytosis
- 3. Energy & Cell Processes
- Introduction to Energy
- Laws of Thermodynamics
- Chemical Reactions
- ATP
- Enzymes
- Enzyme Activation Energy
- Enzyme Binding Factors
- Enzyme Inhibition
- Introduction to Metabolism
- Redox Reactions
- Introduction to Cellular Respiration
- Types of Phosphorylation
- Glycolysis
- Pyruvate Oxidation
- Krebs Cycle
- Electron Transport Chain
- Chemiosmosis
- Review of Aerobic Cellular Respiration
- Fermentation & Anaerobic Respiration
- Introduction to Cell Division
- Organization of DNA in the Cell
- Introduction to the Cell Cycle
- Interphase
- Phases of Mitosis
- Cytokinesis
- Cell Cycle Regulation
- Review of the Cell Cycle
- Cancer
- Introduction to DNA Replication
- DNA Repair
- Central Dogma
- Introduction to Transcription
- Steps of Transcription
- Genetic Code
- Introduction to Translation
- Steps of Translation
- Post-Translational Modification
- 4. Tissues & Histology
- Introduction to Tissues & Histology
- Introduction to Epithelial Tissue
- Characteristics of Epithelial Tissue
- Structural Naming of Epithelial Tissue
- Simple Epithelial Tissues
- Stratified Epithelial Tissues
- Identifying Types of Epithelial Tissue
- Glandular Epithelial Tissue
- Introduction to Connective Tissue
- Classes of Connective Tissue
- Introduction to Connective Tissue Proper
- Connective Tissue Proper: Loose Connective Tissue
- Connective Tissue Proper: Dense Connective Tissue
- Specialized Connective Tissue: Cartilage
- Specialized Connective Tissue: Bone
- Specialized Connective Tissue: Blood
- Introduction to Muscle Tissue
- Types of Muscle Tissue
- Introduction to Nervous Tissue
- Nervous Tissue: The Neuron
- 5. Integumentary System
- 6. Bones & Skeletal Tissue
- An Introduction to Bone and Skeletal Tissue
- Gross Anatomy of Bone: Compact and Spongy Bone
- Gross Anatomy of Bone: Periosteum and Endosteum
- Gross Anatomy of Bone: Bone Marrow
- Gross Anatomy of Bone: Short, Flat, and Irregular Bones
- Gross Anatomy of Bones - Structure of a Long Bone
- Microscopic Anatomy of Bones - Bone Matrix
- Microscopic Anatomy of Bones - Bone Cells
- Microscopic Anatomy of Bones - The Osteon
- Microscopic Anatomy of Bones - Trabeculae
- 7. The Skeletal System
- 8. Joints
- 9. Muscle Tissue
- 10. Muscles
- 11. Nervous Tissue and Nervous System
- 12. The Central Nervous System
- 13. The Peripheral Nervous System
- 14. The Autonomic Nervous System
- 15. The Special Senses
- 16. The Endocrine System
- 17. The Blood
- 18. The Heart
- 19. The Blood Vessels
- 20. The Lymphatic System
- 21. The Immune System
- Introduction to the Immune System
- Introduction to Innate Immunity
- Introduction to First-Line Defenses
- Physical Barriers in First-Line Defenses: Skin
- Physical Barriers in First-Line Defenses: Mucous Membrane
- First-Line Defenses: Chemical Barriers
- First-Line Defenses: Normal Microbiota
- Introduction to Cells of the Immune System
- Cells of the Immune System: Granulocytes
- Cells of the Immune System: Agranulocytes
- Introduction to Cell Communication
- Cell Communication: Surface Receptors & Adhesion Molecules
- Cell Communication: Cytokines
- Pattern Recognition Receptors (PRRs)
- Introduction to the Complement System
- Activation Pathways of the Complement System
- Effects of the Complement System
- Review of the Complement System
- Phagocytosis
- Introduction to Inflammation
- Steps of the Inflammatory Response
- Fever
- Interferon Response
- Review Map of Innate Immunity
- Introduction to Adaptive Immunity
- Antigens
- Introduction to T Lymphocytes
- Major Histocompatibility Complex Molecules
- Activation of T Lymphocytes
- Functions of T Lymphocytes
- Review of Cytotoxic vs Helper T Cells
- Introduction to B Lymphocytes
- Antibodies
- Classes of Antibodies
- Outcomes of Antibody Binding to Antigen
- T Dependent & T Independent Antigens
- Clonal Selection
- Antibody Class Switching
- Affinity Maturation
- Primary and Secondary Response of Adaptive Immunity
- Immune Tolerance
- Regulatory T Cells
- Natural Killer Cells
- Review of Adaptive Immunity
- 22. The Respiratory System
- 23. The Digestive System
- 24. Metabolism and Nutrition
- Essential Amino Acids
- Lipid Vitamins
- Cellular Respiration: Redox Reactions
- Introduction to Cellular Respiration
- Cellular Respiration: Types of Phosphorylation
- Cellular Respiration: Glycolysis
- Cellular Respiration: Pyruvate Oxidation
- Cellular Respiration: Krebs Cycle
- Cellular Respiration: Electron Transport Chain
- Cellular Respiration: Chemiosmosis
- Review of Aerobic Cellular Respiration
- Fermentation & Anaerobic Respiration
- Gluconeogenesis
- Fatty Acid Oxidation
- Amino Acid Oxidation
- 25. The Urinary System
- 26. Fluid and Electrolyte Balance, Acid Base Balance
- 27. The Reproductive System
- 28. Human Development
- 29. Heredity
2. Cell Chemistry & Cell Components
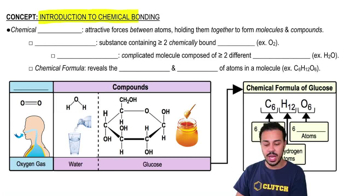
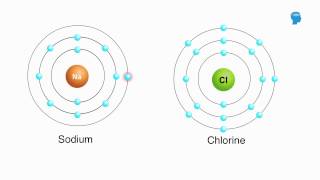
Introduction to Chemical Bonding
2. Cell Chemistry & Cell Components
Introduction to Chemical Bonding
+3
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Open Question
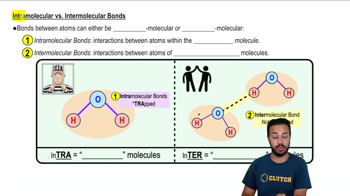
Appropriately label all of the chemical bonds in this image as either intramolecular or intermolecular.
160views - Multiple Choice
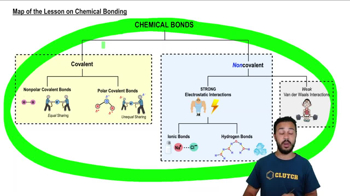
Map of the Lesson on Chemical Bonding
According to the map above, which of the following are types of covalent bonds?
17views - Multiple ChoiceHow would you respond to this reasoning? Oxygen is not a greenhouse gas; therefore, gases containing oxygen—such as ozone, nitrous oxide, and carbon dioxide—are not greenhouse gases either.4views
- Multiple ChoiceAn atom that normally has __________ in its outer shell would not tend to form chemical bonds with other atoms.6views
- Multiple ChoiceThe chemical characteristics or reactivity of an element depend mostly on the __________.3views