Textbook Question
Name the body fluid compartments, noting their locations and the approximate fluid volume in each.
1
views

 Verified step by step guidance
Verified step by step guidance

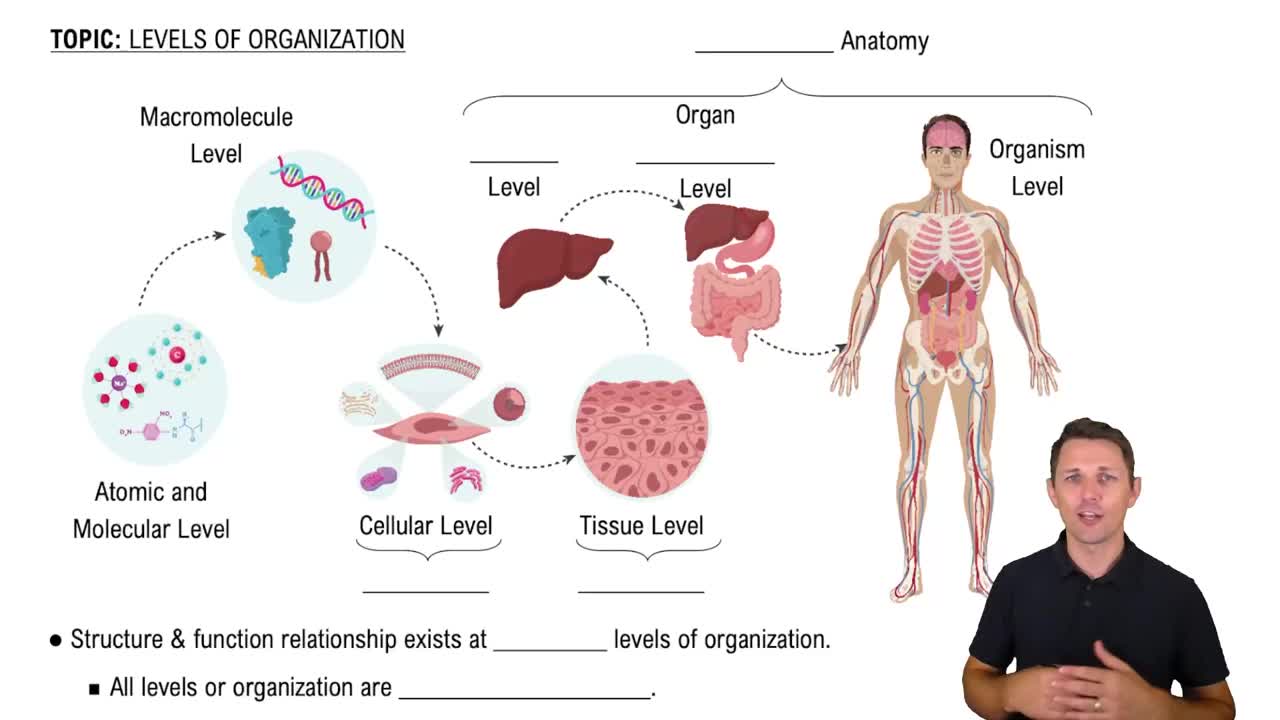
Name the body fluid compartments, noting their locations and the approximate fluid volume in each.
Substance regulated by ADH's effects on the renal tubules is:
a. Ammonium ions
b. Bicarbonate
c. Calcium
d. Chloride
e. Hydrogen ions
f. Magnesium
g. Phosphate
h. Potassium
i. Sodium
j. Water
Describe the thirst mechanism, indicating how it is triggered and terminated.
Explain why and how ECF osmolality is maintained.
Two ions produced during catabolism of glutamine are:
a. Ammonium ions
b. Bicarbonate
c. Calcium
d. Chloride
e. Hydrogen ions
f. Magnesium
g. Phosphate
h. Potassium
i. Sodium
j. Water
Which of the following factors will enhance ADH release?
a. Increase in ECF volume
b. Decrease in ECF volume
c. Decrease in ECF osmolality
d. Increase in ECF osmolality