 Back
BackAnalytical Chemistry: Tools of the Trade – Safety, Measurement, and Laboratory Techniques
Study Guide - Smart Notes

Tools of the Trade
Safety and Chemical Labels
Chemical safety is a foundational aspect of analytical chemistry laboratories. Proper labeling and understanding of chemical hazards are essential for safe handling, storage, and disposal of chemicals. The Occupational Safety and Health Administration (OSHA) provides guidelines for constructing accurate and informative chemical hazard labels.
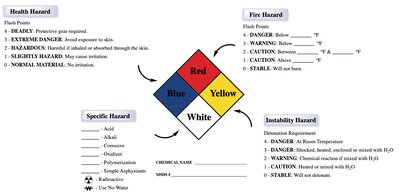
Hazard Diamond: The NFPA (National Fire Protection Association) hazard diamond is a standardized label that communicates health, fire, instability, and specific hazards of chemicals.
Color Codes: Red (fire hazard), Blue (health hazard), Yellow (instability hazard), White (specific hazard).
Numerical Ratings: Each hazard is rated from 0 (minimal) to 4 (severe).
Specific Hazards: Indicated in the white section, such as acid, alkali, corrosive, oxidizer, and water reactivity.
Buoyancy in Analytical Measurements
Buoyancy is the upward force exerted on an object in a fluid (liquid or gas). In analytical chemistry, buoyancy corrections are necessary when weighing substances to obtain accurate mass measurements, as air buoyancy can affect the apparent mass.
Buoyant Force Equation: The force is equal to the weight of the fluid displaced by the object.
Equation:
F: Buoyant force
m: Mass of liquid
g: Gravitational field strength
\rho: Density of liquid
V: Volume of liquid displaced

Example: When calibrating pipets, the mass of water delivered is weighed, and the density of water at the given temperature is used to determine the true volume delivered, accounting for buoyancy effects.
Thermal Dependency in Measurements
Temperature affects the density and volume of liquids, which in turn impacts the accuracy of volumetric and gravimetric measurements. Corrections for thermal expansion are necessary for precise analytical work.
Density of Water: Changes with temperature, requiring reference tables for accurate conversions.
Correction Formula: To correct concentrations or volumes for temperature changes:
c: Original concentration
c': Corrected concentration
d: Original density
d': New density
Example: If a solution is prepared at 21°C and later measured at 26°C, the change in water density must be considered to determine the new concentration.
Laboratory Materials and Apparatus
Analytical chemistry relies on a variety of laboratory apparatuses for measuring, transferring, and handling liquids and solids. Familiarity with these tools is essential for accurate experimental work.
Measuring and Transferring Liquids: Includes pipets, burets, graduated cylinders, and volumetric flasks.
Handling Solids: Includes beakers, Erlenmeyer flasks, crucibles, and evaporating dishes.
Example: To measure exactly 25.0 mL of a solution, a transfer pipet or buret is preferred for accuracy.




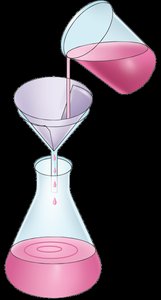
Mixture Separation Techniques: Filtration and Evaporation
Separation of mixtures is a fundamental laboratory skill. Filtration and evaporation are two common techniques used to separate components based on physical properties.
Filtration
Definition: Separation of a solid from a liquid by passing the mixture through a filter.
Residue: The solid left on the filter paper.
Filtrate: The liquid that passes through the filter paper.
Example: Using a coffee filter to separate coffee grounds from brewed coffee.
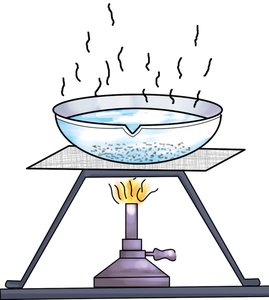
Evaporation
Definition: Separation of a dissolved solid from a liquid by heating the mixture until the liquid evaporates, leaving the solid behind.
Application: Used to recover solutes from solutions or to concentrate solutions.
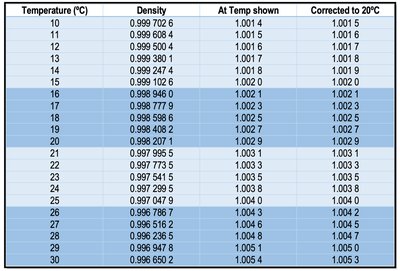
Reference Table: Density of Water at Various Temperatures
The following table provides the density of water at different temperatures, which is essential for correcting volumetric measurements in analytical chemistry.
Temperature (°C) | Density (g/mL) | At Temp shown | Corrected to 20°C |
|---|---|---|---|
10 | 0.999 702 6 | 1.001 4 | 1.001 5 |
15 | 0.999 102 6 | 1.001 9 | 1.002 0 |
20 | 0.998 207 1 | 1.002 8 | 1.002 9 |
25 | 0.997 047 9 | 1.004 3 | 1.004 3 |
30 | 0.996 650 2 | 1.005 4 | 1.005 3 |
