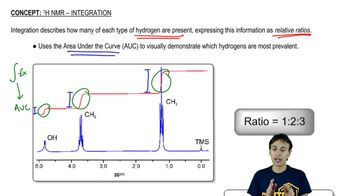
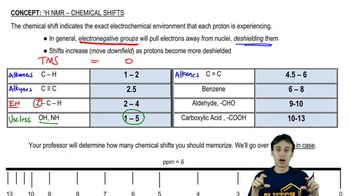
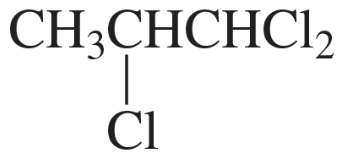Integration and Multiplicity
Integration in 1H NMR refers to the area under each peak, which correlates to the number of hydrogen atoms contributing to that signal. Multiplicity indicates the splitting pattern of the peaks, which arises from neighboring hydrogen atoms (n+1 rule). For example, a singlet indicates no neighboring hydrogens, while a doublet or triplet suggests one or two neighboring hydrogens, respectively. Together, integration and multiplicity provide insights into the hydrogen environment and connectivity within the molecule.