Textbook Question
Predict whether He2+ exists.
2
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1)
Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1) Problem 38(1)
Problem 38(1) Verified step by step guidance
Verified step by step guidance
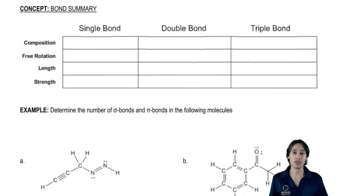


Predict whether He2+ exists.
a. Which bond would be longer?
b. Which bond would be stronger?
3. H—Cl or H—F
Draw the following orbitals:
a. 3s orbital
b. 4s orbital
c. 3p orbital
a. Which bond would be longer?
b. Which bond would be stronger?
2. C—C or C—Cl
Indicate the kind of molecular orbital (σ, σ*, π, or π*) that results when the two atomic orbitals are combined:
a. <IMAGE>
b. <IMAGE>
Which of the bonds in a carbon–oxygen double bond has more effective orbital–orbital overlap: the σ bond or the π bond?