Textbook Question
Explain why compound A has two stereoisomers but compounds B and C exist as single compounds.
3
views

 Verified step by step guidance
Verified step by step guidance

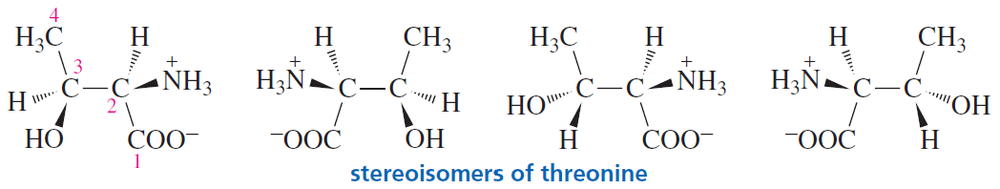
Explain why compound A has two stereoisomers but compounds B and C exist as single compounds.
Chloramphenicol is a broad-spectrum antibiotic that is particularly useful against typhoid fever. What is the configuration of each of its asymmetric centers?
Convert the perspective formula to a skeletal structure.
Convert the Fischer projection to a perspective formula.
Draw a perspective formula for each of the following:
a. (S)-3-chloro-1-pentanol
b. (2R,3R)-2,3-dibromopentane
Draw a perspective formula for each of the following:
c. (2S,3R)-3-methyl-2-pentanol
d. (R)-1,2-dibromobutane