(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(h)

 Mullins 1st Edition
Mullins 1st Edition Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions
Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions Problem 62b
Problem 62b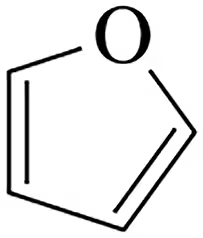
 Verified step by step guidance
Verified step by step guidance

(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(h)
Despite having only sp2-hybridized carbons and having 10 electrons (4n + 2) , the annulene shown is not aromatic. Why?
Using a Frost circle, a student drew the molecular orbital picture shown for the cyclopentadienyl anion, determining it to be antiaromatic. Do you agree with this conclusion? Why or why not?
Use a Frost circle diagram to construct the molecular orbital diagram for the molecules shown. Would you expect them to be aromatic or antiaromatic?
(a)
Use a Frost circle diagram to construct the molecular orbital diagram for the molecules shown. Would you expect them to be aromatic or antiaromatic?
(c)