Textbook Question
Predict whether He2+ exists.
2
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1)
Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1) Problem 26a,b
Problem 26a,b Verified step by step guidance
Verified step by step guidance
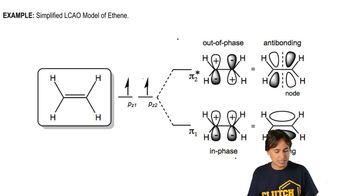
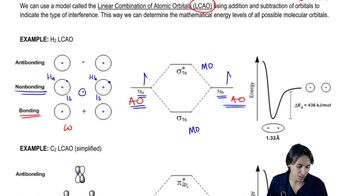
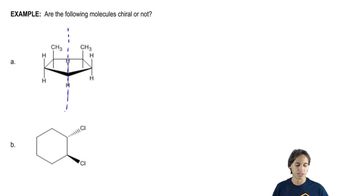
Predict whether He2+ exists.
a. Which bond would be longer?
b. Which bond would be stronger?
3. H—Cl or H—F
Draw the following orbitals:
a. 3s orbital
b. 4s orbital
c. 3p orbital
a. Which bond would be longer?
b. Which bond would be stronger?
2. C—C or C—Cl
Convert the models in Problem 20 to skeletal structures.
a. <IMAGE>
b. <IMAGE>
c. <IMAGE>
d. <IMAGE>
a. Which bond would be longer?
b. Which bond would be stronger?
1. C—Cl or C—I