Draw a Lewis structure, and classify each of the following compounds. The possible classifications are as follows:
alcohol
ether
ketone
aldehyde
carboxylic acid
alkene
(f)
(g)

 Verified step by step guidance
Verified step by step guidance
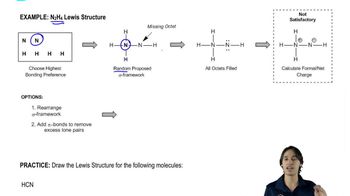
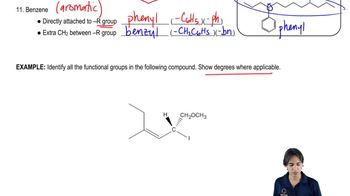
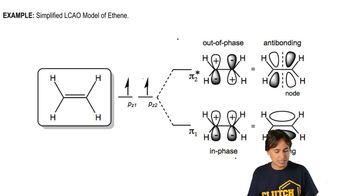
Draw a Lewis structure, and classify each of the following compounds. The possible classifications are as follows:
alcohol
ether
ketone
aldehyde
carboxylic acid
alkene
(f)
(g)
Draw a Lewis structure, and classify each of the following compounds. The possible classifications are as follows: alcohol ether ketone aldehyde carboxylic acid alkene
(d) CH3CH2OCHCH2
(e)
Draw a Lewis structure, and classify each of the following compounds. The possible classifications are as follows:
alcohol
ether
ketone
aldehyde
carboxylic acid
alkene
(h)
(i)
Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into more than one of the following classifications:
alkane
alkene
alkyne
cycloalkane
cycloalkene
cycloalkyne
aromatic
hydrocarbon
(g)
(h)
(i)
Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into more than one of the following classifications:
alkane
alkene
alkyne
cycloalkane
cycloalkene
cycloalkyne
aromatic
hydrocarbon
(d)
(e)
(f)
Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into more than one of the following classifications:
alkane
alkene
alkyne
cycloalkane
cycloalkene
cycloalkyne
aromatic
hydrocarbon
a. (CH3CH2)2CHCH(CH3)2
b. CH3CHCHCH2CH3
c.. CH3CCCH2CH2CH3