Textbook Question
Propose mechanisms for the three imine-forming reactions just shown.
1
views

 Verified step by step guidance
Verified step by step guidance
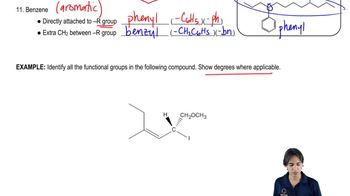
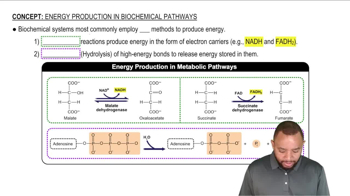
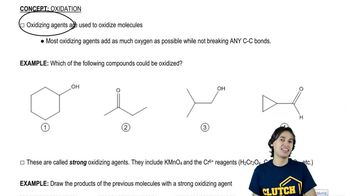
Propose mechanisms for the three imine-forming reactions just shown.
Show how you would accomplish the following syntheses.
c. hexan-1-ol → 2-hydroxyheptanoic acid
Propose a mechanism for each cyanohydrin synthesis just shown.
Show how you would accomplish the following syntheses.
(a) acetophenone → acetophenone cyanohydrin
Propose mechanisms for
(b) the base-catalyzed hydration of acetone to form acetone hydrate.
Depending on the reaction conditions, two different imines of formula C8H9N might be formed by the reaction of benzaldehyde with methylamine. Explain, and give the structures of the two imines.