Textbook Question
Draw the important resonance contributors for the following cations, anions, and radicals.
(b)
8
views

 Wade 9th Edition
Wade 9th Edition Ch. 15 - Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy
Ch. 15 - Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy Problem 26b
Problem 26b Verified step by step guidance
Verified step by step guidance

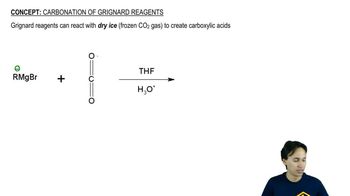
Draw the important resonance contributors for the following cations, anions, and radicals.
(b)
Predict the products of the following reactions.
(h) cyclopentadiene + methyl acrylate, CH2=CH–COOCH3
Show how the reaction of an allylic halide with a Grignard reagent might be used to synthesize the following hydrocarbons.
c. 1-cyclopentylpent-2-ene
Show how the reaction of an allylic halide with a Grignard reagent might be used to synthesize the following hydrocarbons.
a. 5-methylhex-1-ene
Draw the important resonance contributors for the following cations, anions, and radicals.
(a)
Predict the products of the following reactions.
(i)