Conformational studies on ethane-1,2-diol (HOCH2–CH2OH) have shown the most stable conformation about the central C―C bond to be the gauche conformation, which is 9.6 kJ/mol (2.3 kcal/mol) more stable than the anti conformation. Draw Newman projections of these conformers, and explain this curious result.
Ch.3 - Structure and Stereochemistry of Alkanes

Chapter 3, Problem 52a
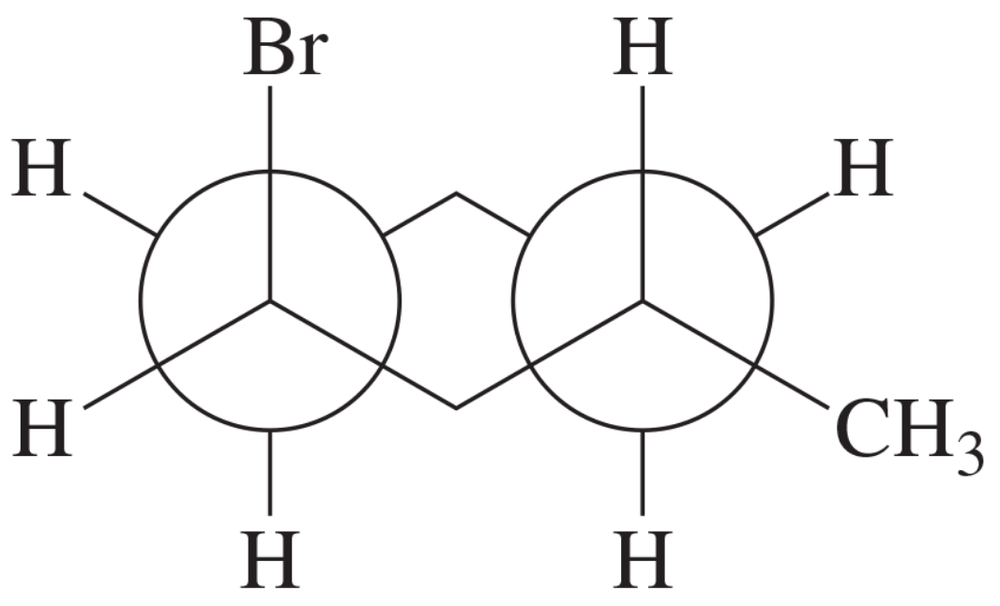
This is a Newman projection of a substituted cyclohexane. a. Draw the equivalent chair form.
 Verified step by step guidance
Verified step by step guidance1
Identify the substituents and their positions in the Newman projection. Note the axial and equatorial positions relative to the cyclohexane ring.
Understand the basic structure of a cyclohexane chair conformation, which alternates between axial and equatorial positions for substituents.
Translate the substituents from the Newman projection to the chair conformation. Ensure that substituents that are axial in the Newman projection remain axial in the chair form, and similarly for equatorial substituents.
Draw the cyclohexane ring in the chair conformation, which resembles a 'zig-zag' pattern. Place the substituents at the correct carbon atoms, maintaining their axial or equatorial orientation.
Double-check the stability of the chair conformation. If necessary, perform a ring flip to see if a more stable conformation can be achieved, where larger substituents are in the equatorial position to minimize steric strain.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
2mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Newman Projection
A Newman projection is a method used in organic chemistry to visualize the conformation of a molecule from a specific angle, typically along a carbon-carbon bond axis. It helps in understanding the spatial arrangement of substituents and their steric interactions, which is crucial for analyzing conformational stability.
Recommended video:
Guided course

Introduction to Drawing Newman Projections
Cyclohexane Conformations
Cyclohexane can adopt several conformations, with the chair form being the most stable due to minimized steric strain and torsional strain. Understanding these conformations is essential for converting a Newman projection into a chair form, as it involves recognizing the spatial arrangement of substituents in three-dimensional space.
Recommended video:
Guided course

Understanding what a conformer is.
Substituted Cyclohexane
Substituted cyclohexanes have additional groups attached to the cyclohexane ring, affecting its conformational preferences. The position of these substituents (axial or equatorial) in the chair conformation influences the molecule's stability and reactivity, making it important to accurately depict these in the equivalent chair form.
Recommended video:
Guided course
Nucleophiles and Electrophiles can react in Substitution Reactions.
Related Practice
Textbook Question
Textbook Question
The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. Draw this most stable conformation of glucose.
4
views
Textbook Question
This is a Newman projection of a substituted cyclohexane.
b. Draw the equivalent structure using wedge and dash notation on a cyclohexane hexagon.
1
views
Textbook Question
Draw Newman projections along the C3―C4 bond to show the most stable and least stable conformations of 3-ethyl-2,4,4-trimethylheptane.
2
views
Textbook Question
This is a Newman projection of a substituted cyclohexane.
c. Give the IUPAC name.
2
views
