Textbook Question
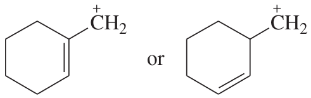
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor.
(c)
1
views

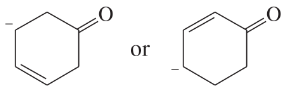
 Verified step by step guidance
Verified step by step guidance
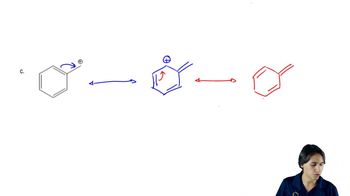

For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor.
(c)
Use resonance structures to identify the areas of high and low electron density in the following compounds:
(a)
(b)
(c)
Use resonance structures to identify the areas of high and low electron density in the following compounds:
g.
h.
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor.
(a)
Use resonance structures to identify the areas of high and low electron density in the following compounds:
i.
j.
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor.
(b)