Textbook Question
Give the systematic names for all alkanes with molecular formula C7H16 that do not have any secondary hydrogens.
1
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 3 - An Introduction to Organic Compounds:Nomenclature, Physical Properties, and Structure
Ch. 3 - An Introduction to Organic Compounds:Nomenclature, Physical Properties, and Structure Problem 70b
Problem 70b Verified step by step guidance
Verified step by step guidance
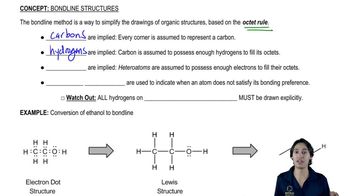

Give the systematic names for all alkanes with molecular formula C7H16 that do not have any secondary hydrogens.
Draw skeletal structures for the following:
c. 2,3,3,4-tetramethylheptane
Draw skeletal structures for the following:
e. 2-methyl-4-(1-methylethyl)octane
Draw skeletal structures for the following:
d. propylcyclopentane
Draw skeletal structures for the following:
a. 5-ethyl-2-methyloctane
Which of the following conformers has the highest energy (is the least stable)?