Textbook Question
d. Is the reaction of 1-butene with HBr regioselective?
e. Is it stereoselective?
f. Is it stereospecific?
1
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 34c
Problem 34c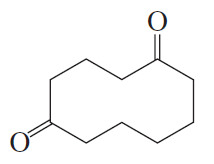
 Verified step by step guidance
Verified step by step guidance
d. Is the reaction of 1-butene with HBr regioselective?
e. Is it stereoselective?
f. Is it stereospecific?
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfide. What is the structure of the alkene?
b.
What characteristics must the reactant of a stereospecific reaction have?
The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfide. What is the structure of the alkene?
a.
The following products were obtained from the oxidative cleavage of a diene. What is the structure of the diene?